Medication-Assisted Treatment Improves Outcomes for Patients With Opioid Use Disorder

This fact sheet has been updated. The latest information is available here.
Overview
Opioid overdoses cause one death every 20 minutes.1 Medication-assisted treatment (MAT)—a combination of psychosocial therapy and U.S. Food and Drug Administration-approved medication—is the most effective intervention to treat opioid use disorder (OUD) and is more effective than either behavioral interventions or medication alone.2 MAT significantly reduces illicit opioid use compared with nondrug approaches,3 and increased access to these therapies can reduce overdose fatalities.4 However, MAT is often unavailable to those in need of it because of inadequate funding for treatment programs and a lack of qualified providers who can deliver these therapies.5
Background
OUD is a chronic brain disease caused by the recurrent use of opioids, including prescription drugs, such as oxycodone and hydrocodone, and illicit substances such as heroin. OUD includes dysfunction of the brain reward system, motivation, memory, and related circuitry and is reflected in individuals “pathologically pursuing reward and/or relief by substance use and other behaviors.”6 As with other chronic relapsing conditions, the clinical course of OUD includes periods of exacerbation and remission, but the patient is never disease-free.7
What is MAT?
MAT pairs nondrug therapies, such as counseling or cognitive behavioral therapy, with an FDA-approved medication to treat OUD. These drugs—methadone, buprenorphine, and naltrexone—are available in various product formulations and doses. Each medication differs in the way it works to relieve symptoms of opioid withdrawal and/or block the euphoric effects of the drugs. Consistent with the approach used for other chronic diseases such as diabetes, treatment plans for OUD are patient specific and created with input from the patient, the prescriber, and other members of the health care team. This dual approach to OUD treatment is supported by medical and behavioral health groups, including the American Society of Addiction Medicine and the National Council for Behavioral Health, patient advocate groups, and federal entities including the White House Office of National Drug Control Policy and the U.S. surgeon general.8
Medication-assisted treatment saves lives while increasing the chances a person will remain in treatment and learn the skills and build the networks necessary for long-term recovery.Michael Botticelli, director, national drug control policy
Psychosocial treatment
Psychosocial treatment, also known as behavioral health treatment, is recommended in conjunction with all drug therapies for OUD. Patients receiving psychosocial treatment have better outcomes than patients who do not.9 Common therapeutic goals of psychosocial treatment are to:
- Modify underlying behaviors that may lead patients to misuse opioids.
- Encourage patients to adhere to their prescribed medications.
- Treat any other existing psychiatric disorders.10 Psychosocial treatment begins with an assessment of a patient’s psychosocial needs and the development of a patient-specific treatment plan. Treatment may include one or more of the following:
- Individual or group counseling.
- Connection to family support systems, including family therapy.
- Referrals to community-based services.
- Contingency management—an evidence-based intervention that provides tangible rewards (often vouchers to exchange for retail goods and services) for positive behaviors such as abstaining from opioids.
- Mutual help programs, such as the Narcotics Anonymous 12-step facilitation therapy, may also be offered as an ancillary service.11
FDA-approved medications
Methadone, buprenorphine, and naltrexone are the only FDA-approved medications to treat OUD. Under federal regulations for methadone treatment, the drug must be administered daily in a certified opioid treatment program (OTP).12 In accordance with federal law, buprenorphine may be prescribed on a weekly or monthly basis for at-home use, but clinicians must apply for waivers to be able to prescribe these drugs.13 In contrast, naltrexone can be prescribed by any clinician authorized to dispense medications. These three treatment options have different mechanisms of action. The drug effects—in addition to patient-specific factors, such as response to past treatment—guide therapy selection. These factors are highlighted in Table 1.
Opioid Treatment Program
An OTP is a facility where patients can take medications under the supervision of staff and receive other care services. These programs are regulated and certified by the federal Substance Abuse and Mental Health Services Administration (SAMHSA) and can exist in a number of care settings, including intensive outpatient, residential, and hospital settings. An OTP is the only venue where patients can receive methadone for the treatment of OUD.14
Buprenorphine Waivers
Clinicians who wish to prescribe buprenorphine for the treatment of OUD must qualify for a waiver under the Drug Addiction Treatment Act of 2000. Prescribers who do not have advanced credentials in addiction psychiatry or addiction medicine must complete eight hours of training, available online or in person. Once approved by SAMHSA, prescribers may treat a designated number of patients with approved buprenorphine products in any outpatient practice setting.15
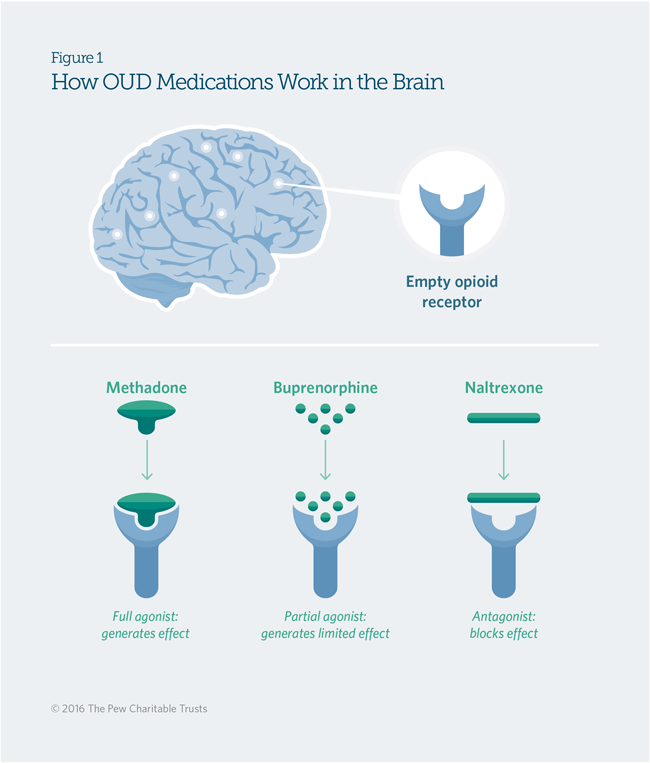
Mechanisms of action
Receptors, which are found on human cells, are places where a drug or substance made by the body binds to either cause or block an effect. Methadone and buprenorphine are opioid agonists, meaning they activate or occupy the mu-opioid receptor, the same one activated by heroin.
Methadone is a full agonist. By fully occupying the mu-opioid receptor, methadone lessens the painful symptoms of opiate withdrawal and blocks the euphoric effects of other opioid drugs. Unlike heroin and other misused opioid agonists, methadone is longer lasting, usually 24 to 36 hours, preventing the frequent peaks and valleys associated with drug-seeking behavior.16 No optimal length of treatment with methadone has been established; however, 12 months is considered a minimum for methadone maintenance.17
Buprenorphine is a partial agonist, meaning it does not completely bind to the mu-opioid receptor. As a result, buprenorphine has a ceiling effect, meaning that its effects will plateau and will not increase even with repeated dosing. Buprenorphine does not produce euphoria and does not have some of the dangerous side effects associated with other opioids.18 The optimal duration of treatment is unknown, and decisions to discontinue treatment with buprenorphine are patient-specific. Once this decision is made, the process of safely tapering the buprenorphine dose often spans many months.19
Naltrexone is an opioid antagonist, meaning that it covers, rather than activates, the mu-opioid receptor, effectively blocking the effects of opioids if they are used. Opioid antagonists do not produce any euphoric effects, so no physical dependence is associated with their use.20 Opioid antagonists are recommended for relapse prevention and for abstinence-based treatment, not for withdrawal management.

Evidence of effectiveness
For most people, the use of medications combined with psychosocial treatment is superior to drug or psychosocial treatment on its own.22 For example, research shows that MAT significantly increases a patient’s adherence to treatment and reduces illicit opioid use compared with nondrug approaches.23 By reducing risk behaviors such as injection of illicit drugs, it also decreases transmission of infectious diseases such as HIV and hepatitis C.24
Treatment gap
Despite MAT’s demonstrated effectiveness, many people are unable to access its benefits. One reason is that treatment programs have been slow to offer MAT drugs. Only 23 percent of publicly funded treatment programs reported offering any FDA-approved medications to treat substance use disorders, and less than half of private-sector treatment programs reported that their physicians prescribed FDA-approved medications.25 Two key barriers to the use of MAT are limited insurance coverage and a lack of qualified medical personnel. Although the Affordable Care Act mandates that insurance companies pay for OUD treatment, it does not specify which benefits must be covered. As a result, drug and behavioral therapies that may be optimal for a specific patient are not always covered. In addition, treatment services may be covered only for a specific period of time, creating harmful limitations.26 It is particularly important that Medicaid cover a broad range of treatment options because of its influence on available program services; for example, Medicaid coverage for buprenorphine is a significant predictor of its adoption by community-based treatment programs.27 The lack of authorized buprenorphine prescribers for treatment of OUD is also a critical factor in the treatment gap. Across the country, the number of people with opioid dependence far exceeds the number of eligible providers able to prescribe buprenorphine for OUD.28 In some areas, this gap is exacerbated by geography: 30 million Americans live in counties that do not have any physicians with waivers allowing them to prescribe buprenorphine for MAT.29
Curbing opioid use disorders with MAT
MAT is the most effective treatment for OUD. The individual treatment plan may include multiple types of cognitive therapies, and patients may need to try more than one drug to achieve goals. Improving awareness of how MAT works, ensuring comprehensive coverage of all services, and expanding access to eligible providers will be integral factors in curbing the opioid epidemic.
Endnotes
- Rose A. Rudd et al., “Increases in Drug and Opioid Overdose Deaths—United States, 2000-2014,” Morbidity and Mortality Weekly Report 64, no. 50 (2016): 1378–82, http://www.cdc.gov/mmwr/preview/mmwrhtml/mm6450a3.htm?s_cid=mm6450a3_w.
- American Society of Addiction Medicine, The ASAM National Practice Guideline for the Use of Medications in the Treatment of Addiction Involving Opioid Use (2015), http://www.asam.org/docs/default-source/practice-support/guidelines-and-consensus-docs/asam-national-practice-guideline-supplement.pdf?sfvrsn=24; and U.S. Department of Health and Human Services, Addressing Prescription Drug Abuse in the United States: Current Activities and Future Opportunities” (2013), https://www.cdc.gov/drugoverdose/pdf/ hhs_prescription_drug_abuse_report_09.2013.pdf.
- Richard P. Mattick et al., “Methadone Maintenance Therapy Versus No Opioid Replacement Therapy for Opioid Dependence,” Cochrane Database of Systematic Reviews 3 (2009): CD002209, http://www.ncbi.nlm.nih.gov/pubmed/19588333; Sandra D. Comer et al., “Injectable, Sustained-Release Naltrexone for the Treatment of Opioid Dependence: A Randomized, Placebo-Controlled Trial,” Archives of General Psychiatry 63, no. 2 (2006): 210–8, http://archpsyc.jamanetwork.com/article.aspx?articleid=209312; and Paul J. Fudala et al., “Office-Based Treatment of Opiate Addiction With a Sublingual-Tablet Formulation of Buprenorphine and Naloxone,” New England Journal of Medicine 349, no. 10 (2003): 949–58, http://www.ncbi.nlm.nih.gov/pubmed/12954743.
- Robert P. Schwartz et al., “Opioid Agonist Treatments and Heroin Overdose Deaths in Baltimore, Maryland, 1995-2009,” American Journal of Public Health 103, no. 5 (2013): 917–22, http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3670653.
- Hannah K. Knudsen, Paul M. Roman, and Carrie B. Oser, “Facilitating Factors and Barriers to the Use of Medications in Publicly Funded Addiction Treatment Organizations,” Journal of Addiction Medicine 4, no. 2 (2010): 99–107, https://www.ncbi.nlm.nih.gov/pubmed/20835350.
- American Society of Addiction Medicine, The ASAM National Practice Guideline.
- Charles P. O’Brien, “Drug Addiction,” in Goodman & Gilman’s the Pharmacological Basis of Therapeutics, eds. Laurence L. Brunton, Bruce A. Chabner, and Björn C. Knollmann, 12th ed. (New York: McGraw Hill, 2011), 649-66, http://accessmedicine.mhmedical.com/content.aspx?bookid=1613§ionid=102159667.
- Substance Abuse and Mental Health Services Administration, “Medication-Assisted Treatment (MAT) Support Organizations,” last modified Sept. 25, 2015, http://www.samhsa.gov/medication-assisted-treatment/training-resources/support-organizations; and National Council for Behavioral Health, letter to SAMHSA, May 26, 2016, http://www.thenationalcouncil.org/wp-content/uploads/2016/05/NPRM-Medication-Assisted-Treatment-2.pdf.
- Lissa Dutra et al., “A Meta-Analytic Review of Psychosocial Interventions for Substance Use Disorders,” American Journal of Psychiatry 165, no. 2 (2008): 179-87, https://www.researchgate.net/publication/5653816_A_Meta-Analytic_Review_of_Psychosocial_Interventions_for_Substance_Use_Disorders.
- American Society of Addiction Medicine, The ASAM National Practice Guideline.
- Ibid.; and National Institute on Drug Abuse, Principles of Drug Addiction Treatment: A Research-Based Guide (2012), https://d14rmgtrwzf5a.cloudfront.net/sites/default/files/podat_1.pdf.
- American Society of Addiction Medicine, The ASAM National Practice Guideline.
- Drug Enforcement Administration, “DEA Requirements for DATA Waived Physicians (DWPs),” https://www.deadiversion.usdoj.gov/pubs/docs/dwp_buprenorphine.htm; and American Society of Addiction Medicine, The ASAM National Practice Guideline.
- Center for Substance Abuse Treatment, Medication-Assisted Treatment for Opioid Addiction in Opioid Treatment Programs: Inservice Training (2009), https://store.samhsa.gov/shin/content/SMA09-4341/SMA09-4341.pdf.
- Definition is slightly modified based on changes to legislation. American Society of Addiction Medicine, The ASAM National Practice Guideline.
- Ibid.
- National Institute on Drug Abuse, Principles of Drug Addiction Treatment.
- American Society of Addiction Medicine, The ASAM National Practice Guideline.
- Ibid.
- Marc A. Schuckit, “Treatment of Opioid-Use Disorders,” New England Journal of Medicine 375, no. 4 (2016): 357–68, http://dx.doi.org/10.1056/NEJMra1604339.
- American Society of Addiction Medicine, The ASAM National Practice Guideline.
- Ibid.
- Mattick et al., “Methadone Maintenance Therapy”; Comer et al., “Injectable, Sustained-Release Naltrexone”; and Fudala et al., “Office-Based Treatment.”
- Schwartz et al., “Opioid Agonist Treatments”; Judith I. Tsui et al., “Association of Opioid Agonist Therapy With Lower Incidence of Hepatitis C Virus Infection in Young Adult Injection Drug Users,” JAMA Internal Medicine 174, no. 12 (2014): 1974–81, http://archinte.jamanetwork.com/article.aspx?articleid=1918926; and David S. Metzger et al., “Human Immunodeficiency Virus Seroconversion Among Intravenous Drug Users In- and Out-of-Treatment: An 18-Month Prospective Follow-Up,” Journal of Acquired Immune Deficiency Syndromes 6, no. 9 (1993): 1049–56, http://www.ncbi.nlm.nih.gov/pubmed/8340896.
- Knudsen, Roman, and Oser, “Facilitating Factors and Barriers”; Hannah K. Knudsen, Amanda J. Abraham, and Paul M. Roman, “Adoption and Implementation of Medications in Addiction Treatment Programs,” Journal of Addiction Medicine 5, no. 1 (2011): 21–7, http://www.ncbi.nlm.nih.gov/pubmed/21359109.
- National Center on Addiction and Substance Abuse, Uncovering Coverage Gaps: A Review of Addiction Benefits in ACA Plans (June 2016), http://www.centeronaddiction.org/addiction-research/reports/uncovering-coverage-gaps-review-of-addictionbenefits-in-aca-plans.
- Lori J. Ducharme and Amanda J. Abraham, “State Policy Influence on the Early Diffusion of Buprenorphine in Community Treatment Programs,” Substance Abuse Treatment Prevention Policy 3, no. 17 (2008), http://dx.doi.org/10.1186/1747-597X-3-17.
- Christopher M. Jones et al., “National and State Treatment Need and Capacity for Opioid Agonist Medication-Assisted Treatment,” American Journal of Public Health 105, no. 8 (2015): e55–63, http://dx.doi.org/10.2105/AJPH.2015.302664.
- Roger A. Rosenblatt et al., “Geographic and Specialty Distribution of US Physicians Trained to Treat Opioid Use Disorder,” Annals of Family Medicine 13, no. 1 (2015): 23–6, http://dx.doi.org/10.1370/afm.1735.


America’s Overdose Crisis
Sign up for our five-email course explaining the overdose crisis in America, the state of treatment access, and ways to improve care
Sign up










