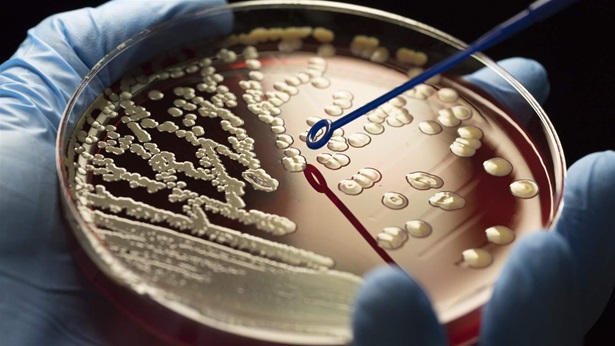
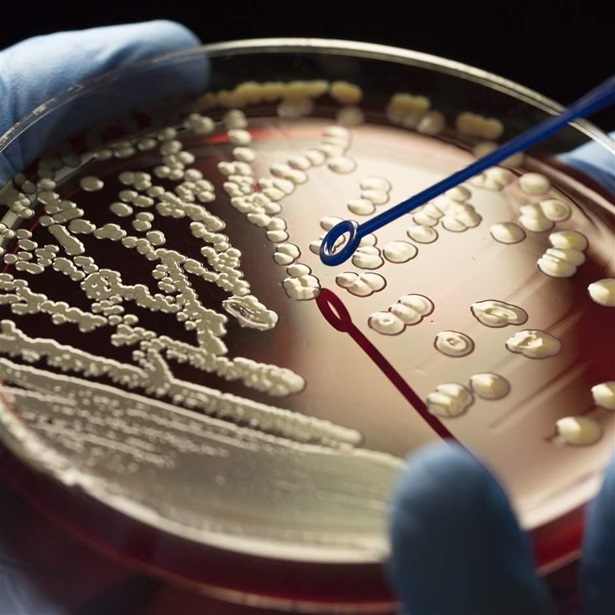
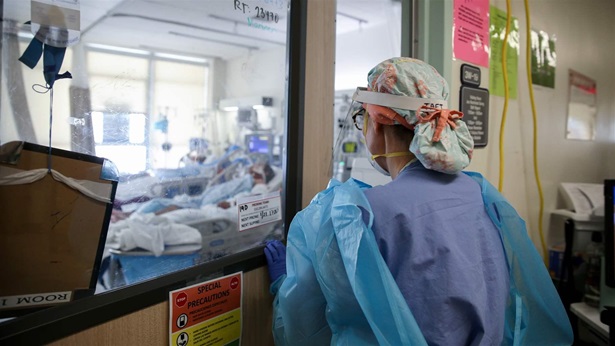
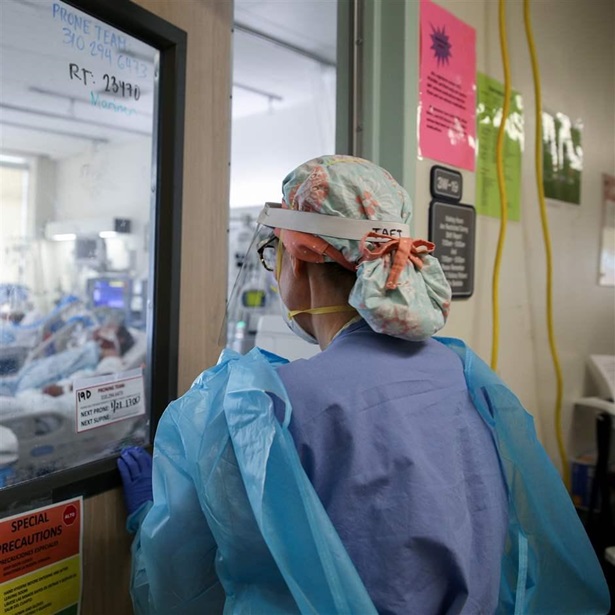
Antibiotics are fundamental to modern medicine, essential for treating everything from routine skin infections to strep throat, and for protecting vulnerable patients receiving chemotherapy or being treated in intensive care units.
Although antibiotic resistance is not a new problem, its scope now constitutes a major threat to human health. According to the Centers for Disease Control and Prevention, Americans contract more than 2.8 million antibiotic-resistant infections every year—and at least 35,000 die.
Medical and public health experts agree that addressing antibiotic resistance requires measures that will ensure both the prudent use of existing drugs and a suite of economic incentives to spur urgently needed antibiotic innovation.
Project Goals
Over the years, Pew has addressed a wide range of challenges in the antibiotic resistance problem—from reducing the need for antibiotic use in food animals and overcoming scientific barriers to antibiotic innovation to improving the collection and reporting of antibiotic use data in all settings. Today, the antibiotic resistance project is focused on advancing policies that would:
- Spur the creation of new antibiotics by removing the economic obstacles that impede antibiotic discovery and development.
- Establish stewardship programs to ensure that antibiotics are prescribed appropriately in human health care settings.
Our Work
This video is hosted by YouTube. In order to view it, you must consent to the use of “Marketing Cookies” by updating your preferences in the Cookie Settings link below. View on YouTube
This video is hosted by YouTube. In order to view it, you must consent to the use of “Marketing Cookies” by updating your preferences in the Cookie Settings link below. View on YouTube