New Way to Target Lung Tumors Unveiled Through Creative Research
A protein’s role in helping such cancers respond to their environment may be a key to innovative therapies
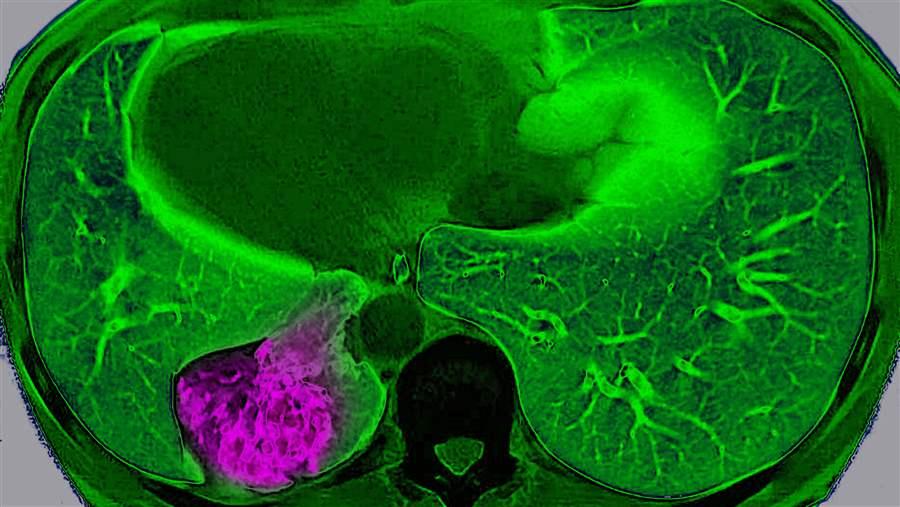
An MRI view from above shows a cancerous tumor (in purple) on a patient’s left lung. New research explores how lung cancer adapts to its environment, which could lead to new treatments.
BSIP/UIG/Getty ImagesLung cancer is the second most common cancer in men and women, making up about 14 percent of all new cancers. On Sunday, we will observe World Cancer Day to raise awareness about the disease. Cancers in the lung are of particular interest to researchers because it is not clear how they adapt to and grow within the lung’s high-oxygen environment—a condition normally toxic to cells.
Richard Possemato, Ph.D., a 2016 Pew-Stewart Scholar for Cancer Research at the Perlmutter Cancer Center at NYU Langone Health, is exploring how cancer cells respond to changes in the tumor’s surroundings and how cellular pathways that control the tumor’s metabolism change to help it survive.
Possemato and 2003 Pew scholar David M. Sabatini, M.D., Ph.D. at the Whitehead Institute for Biomedical Research, both corresponding authors on the study, reported recently in the journal Nature that lung cancer cells are able to thrive in the lung despite the oxygen burden because of increased levels of a protective protein called NFS1.
The protein groups essential elements together to form iron-sulfur clusters (ISCs), which help support the function of numerous proteins needed for processes such as DNA repair, metabolism, and respiration. In oxygen-rich environments such as the lung, ISCs are readily destroyed, and NFS1 plays a crucial role in creating new ISCs.
Possemato and Sabatini found that NFS1 was central to protecting lung cancer cells against high amounts of oxygen, allowing them to thrive and grow in the lung. Notably, NFS1 was not found to be critical for cells’ survival in low-oxygen conditions.
Their study may provide a new approach for destroying lung cancer cells through the inhibition of NFS1. Possemato’s theory that if NFS1 is blocked, cancer cells will quickly run out of ISCs, and cellular processes will shut down and prevent the cell from dividing, is an exciting possibility. Additionally, it’s possible that lack of NFS1 activity may also lead to excess iron buildup and cell death.
In the future, the team plans to test this idea by identifying compounds that block NFS1 and determining whether they can halt tumor development.
Kara Coleman directs The Pew Charitable Trusts’ biomedical programs, including the biomedical scholars, Pew-Stewart Scholars for Cancer Research, and Latin American fellows programs.









