Dietary Supplement Use in the Military
Overview
Many service members take dietary supplements to enhance performance, build muscle, lose weight, or promote general well-being. Taking one or more of these products may be beneficial when a person does not get the recommended amount of nutrients from foods.1 Unfortunately, some supplements can cause serious injury or death. Dangerous supplements compromise the readiness of our armed forces and the health of our soldiers.
Supplement use in the military is common
More U.S. service members take supplements than does the general population.
- About 70 percent of military personnel report using a supplement, compared with about half of civilians.2
- 22 percent of service members consume three or more supplements a week.3
- The most common reasons that soldiers take these products are to promote general health, increase energy, build muscle strength, and enhance performance.4
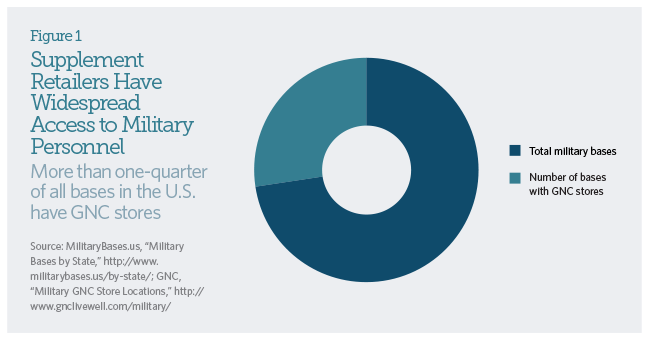
- Dietary supplements are sold on military bases across the country. For example, there are more than 100 GNC stores on military bases in the U.S.5
Supplements have harmed military personnel
Service members have unique occupational demands, including intense cognitive and physical tasks, exposure to high levels of environmental stress, and chronic sleep loss. With these heightened performance requirements, many soldiers turn to supplements to enhance their mental and physical health. But certain supplements can pose a risk.
In 2011, 10 percent of U.S. military personnel took at least one dietary supplement a day that could pose a risk of causing injury to the heart or liver, or another significant adverse reaction.6
Side effects of dietary supplements, particularly protein powders, herbal substitutes for hormones, and combination products, can range from mild (e.g., abnormal heartbeat, tremors, stomach pain, dizziness, and numbness) to serious (e.g., heart attacks, strokes, and death).7
Some supplements contain undeclared drugs, steroids, or stimulants—posing another risk to consumers. Often, these unsafe ingredients are found in the supplements most commonly taken by military personnel.8
Examples
DMAA: In 2011, a 22-year-old Army private in Texas collapsed during a routine exercise and died of cardiac arrest later that day. According to court filings,9 it was later discovered that he had taken a performance-enhancing supplement called Jack3d, bought at a GNC store on the base.10 The supplement contained a stimulant called 1,3-dimethylamylamine (DMAA), which can have potentially harmful side effects. It is an active ingredient in some bodybuilding, weight loss, and performance-enhancing supplements. It can cause high blood pressure and heart attacks.11
As a precaution, the Department of Defense removed all DMAA-containing products from military bases in 2011.12 The following year, the U.S. Food and Drug Administration announced that dietary supplements containing DMAA were illegal because the ingredient has not been shown to be “reasonably expected to be safe.”13 In 2013, a DOD Safety Review Panel reported that four soldiers who died following physical exercise were found to have traces of DMAA in their blood.14 However, while many products have been removed from the market, some retailers still appear to stock old or reformulated products containing the ingredient.15
DMBA: Nearly three years after military bases pulled supplements containing DMAA from their shelves, the DOD banned a similar ingredient for safety concerns. Supplements containing the stimulant 1,3-dimethylbutylamine (DMBA) were removed from stores on military bases because no reliable scientific studies had been conducted on this ingredient’s safety, and its health effects were unknown.16 In 2015, FDA issued warning letters to 14 companies17 manufacturing products with DMBA, alleging that they were marketing adulterated supplements with a new dietary ingredient lacking evidence for safe use.18
SARMs: A new threat to soldiers’ health and military readiness is dietary supplements containing selective androgen receptor modulators (SARMs)—synthetic drugs designed to mimic the effects of testosterone. Depending on the specific chemical, SARMs can be either unapproved new drugs or new dietary ingredients, and without a new dietary ingredient notification, they would be illegal in dietary supplements. The DOD has identified dozens of products containing SARMs, particularly those marketed for bodybuilding.19 In 2014, FDA sent a warning letter to one manufacturer of several products containing SARMs, warning the company that it was allegedly marketing misbranded and potentially harmful dietary supplements that contain an unapproved new drug.20
Caffeine: FDA considers caffeine in coffee, colas, and other foods safe up to a certain limit (400 milligrams a day or about four to five cups of coffee).21 However, the amount of caffeine in dietary supplements is often not listed on the label, and it may be much higher. Dietary supplements with caffeine are marketed as pre-workout or energy supplements to improve mental performance and alertness for people who are sleep-deprived or otherwise fatigued. Caffeine is most commonly listed on dietary supplement ingredient labels as “caffeine anhydrous,” but other names for it include 1,3,7-trimethylxanthine and methyltheobromine.22 Hidden sources of caffeine, including yerba mate, green tea extract, cacao beans, and kola/ cola nuts, are often found in weight-loss and performance-enhancing dietary supplements.23 According to the DOD, high doses of caffeine (above 600 mg/day) can cause serious side effects, including abnormally fast heart rate, tremors, insomnia, and arrhythmia.24 A literature review attributed the deaths of 51 people to the ingestion of toxic concentrations of caffeine, which can only be achieved through consuming it in tablet or powder form.25 In 2014, FDA reported that two young men died after ingesting powdered caffeine, which contained the caffeine equivalent of roughly 25 cups of coffee per teaspoon.26 FDA sent warning letters to five companies27 notifying them that their products pose a significant risk to consumers.28
Supplements can cause false positive drug tests
Taking certain supplements, especially those marketed for bodybuilding and weight loss, may cause soldiers to test positive on military drug tests. This occurs when the supplement contains undeclared or known drug ingredients.29 There is no way to know whether a supplement contains an undeclared drug without testing the product, but the DOD has identified several ingredients that may cause false positive drug tests, including supplements containing steroidlike ingredients such as prohormones, testosterone “boosters,” and “designer steroids,” or anything else compared with or purported to behave as an anabolic steroid.30
How unsafe supplements reach the market
Supplements are not subject to the same regulatory oversight as drugs, which must demonstrate safety and effectiveness before they can be marketed.31 In 1994, Congress passed the Dietary Supplement Health and Education Act, which reaffirmed that dietary supplements are a category of food under the Federal Food, Drug, and Cosmetic Act, and established a framework that is more similar to the regulation of foods than drugs.32 This means that under current law, dietary supplements do not need premarket approval from FDA, and the agency is not notified that they are on the market unless the manufacturer intends to make a truthful and nonmisleading structure/function claim about the product33 or it contains a “new dietary ingredient”—a dietary ingredient that was not on the market in the U.S. before Oct. 15, 1994.34
The degree of quality control for dietary supplements depends on the ingredient suppliers, manufacturers, and others in the production process. Manufacturers must ensure that the identity, purity, strength, and composition of a dietary supplement product are accurate as labeled. If dietary supplements contain contaminants or have an ingredient not listed on the label, FDA considers those products to be adulterated or misbranded, respectively.35 Although FDA inspects registered dietary supplement manufacturers, it is only able to get to a small fraction of them. Not every entity selling dietary supplements is registered with FDA, and even those that are registered are not required to tell FDA what they are making, which makes it impossible for the agency to prioritize inspections based on risk. Owing in part to resource constraints, the agency cannot routinely track compliance with manufacturing regulations. But even its limited number of inspections reveals quality and safety concerns. In 2015, FDA reported that it had inspected 483 of nearly 13,000 registered dietary supplement manufacturing facilities and found violations in 281 of them.36
In 2015, FDA found violations at more than half of the manufacturing facilities it inspected
Steps to improve safety
Educating military personnel may be an effective way to increase awareness within the DOD community about potential risks and how to choose safe dietary supplements. In 2012, the DOD launched such a campaign— Operation Supplement Safety (OPSS)—for all branches of the armed services. As part of the effort, OPSS collaborates with FDA and other government and professional organizations to provide service members with up-to-date information about dietary supplements.37
To more efficiently monitor the dietary supplement marketplace, and take quick action if needed, FDA should have access to more information about it, such as the names of specific products, what is in those products, and how those products are labeled. Manufacturers of dietary supplements should have incentives to provide FDA with that information. FDA also needs more resources to carry out existing enforcement actions. More mechanisms to allow the agency to distinguish quality supplements from problematic ones would help ensure that service members have access to supplements that are safe and of high quality.
Endnotes
- U.S. Department of Agriculture and U.S. Department of Health and Human Services, “Dietary Guidelines for Americans 2015–2020” (2015), https://health.gov/dietaryguidelines/2015/resources/2015-2020_Dietary_Guidelines.pdf.
- Krista G. Austin et al., “Multivitamin and Protein Supplement Use Is Associated With Positive Mood States and Health Behaviors in US Military and Coast Guard Personnel,” Journal of Clinical Psychopharmacology 34, no. 5 (2014): 595–601, http://dx.doi.org/10.1097/JCP.0000000000000193; Elizabeth D. Kantor et al., “Trends in Dietary Supplement Use Among U.S. Adults From 1999–2012,” Journal of the American Medical Association 316, no. 14 (2016): 1464–74, http://dx.doi.org/10.1001/jama.2016.14403.
- Harris Lieberman et al., “Use of Dietary Supplements Among Active-Duty US Army Soldiers,” The American Journal of Clinical Nutrition 92, no. 4 (2010): 985–95, http://dx.doi.org/10.3945/ajcn.2010.29274.
- Ibid.
- GNC, “Military GNC Store Locations,” accessed May 15, 2017, http://www.gnclivewell.com/military/.
- Patricia A. Deuster and Harris R. Lieberman, “Protecting Military Personnel From High Risk Dietary Supplements,” Drug Testing and Analysis 8, no. 3-4 (2015): 431–33, http://dx.doi.org/10.1002/dta.1866.
- Krista G. Austin et al., “Self-Reported Side-Effects Associated With Use of Dietary Supplements in an Armed Forces Population,” Drug Testing and Analysis 8, no. 3-4 (2016): 287–95, http://dx.doi.org/10.1002/dta.1905; David Samenuk et al., “Adverse Cardiovascular Events Temporally Associated With Ma Huang, an Herbal Source of Ephedrine,” Mayo Clinic Proceedings 77, no. 1 (2002): 12–16, http://dx.doi.org/10.4065/77.1.12.
- Deuster and Lieberman, “Protecting Military Personnel.”
- A federal judge ruled on Jan. 27, 2016, to dismiss the wrongful death lawsuit against USPlabs, affirming a magistrate’s earlier decision to strike three experts’ testimonies showing causation between consumption of Jack3d and the Army private’s death. Josh Long, “USPlabs Overcomes Lawsuit Claiming DMAA in Supplement Contributed to Soldier’s Death,” Natural Products Insider, Jan. 29, 2016, https://www.naturalproductsinsider.com/blogs/insider-law/2016/01/usplabs-overcomes-lawsuit-claiming-dmaa-in-supple.aspx.
- Peter Hutt, representation for USPlabs, manufacturer of Jack3d, responded that the company did not have knowledge of any deaths as a result of consumption of its Jack3d product. He also added that there is no causal link between use of the product and the reported deaths. GNC also shared in a statement that it has “no reason to believe DMAA is unsafe.” Natasha Singer and Peter Lattman, “Is the Seller to Blame? Workout Supplement Challenged After Death of Soldier,” The New York Times, March 15, 2013, http://www.nytimes.com/2013/03/17/business/a-soldiers-parents-take-aim-at-gnc-and-a-supplement-maker.html.
- U.S. Food and Drug Administration, “Stimulant Potentially Dangerous to Health, FDA Warns,” last modified Jan. 12, 2016, https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm347270.htm.
- U.S. Department of Defense, “Report of the Department of Defense 1,3 Dimethylamylamine (DMAA) Safety Review Panel” (2013), http://www.webcitation.org/6cZi3FUku.
- U.S. Food and Drug Administration, “DMAA in Dietary Supplements,” last modified Oct. 19, 2015, https://www.fda.gov/Food/DietarySupplements/ProductsIngredients/ucm346576.htm.
- U.S. Department of Defense, “Report of the Department of Defense 1,3 Dimethylamylamine (DMAA) Safety Review Panel.”
- PricePlow, “DMAA Supplement List—What’s Still Available in 2017?” accessed June 7, 2017, https://blog.priceplow.com/dmaa-supplements.
- Human Performance Resource Center, “Is DMBA the Same Thing as DMAA? Why Were These Products Pulled From Stores on Military Base?” accessed May 15, 2017, http://hprc-online.org/dietary-supplements/opss/operation-supplement-safety-OPSS/opss-frequently-asked-questions-faqs-1/is-dmba-the-same-thing-as-dmaa-and-why-was-it-pulled-from-stores-on-military-base.
- U.S. Food and Drug Administration, “DMBA in Dietary Supplements,” last modified April 28, 2015, https://www.fda.gov/Food/DietarySupplements/ProductsIngredients/ucm444719.htm.
- Ibid. Six of the 14 companies submitted corrective actions in response to warning letters received, but as of August 2017, more than half of the companies had not responded to FDA’s warning letter.
- Operation Supplement Safety, “Dietary Supplements and Other Commercial Products Containing SARMs,” accessed May 15, 2017, https://www.opss.org/docs/dietary-supplements-and-other-commercial-products-containing-sarms.
- U.S. Food and Drug Administration, “Biogenix USA, LLC Warning Letter (15-ATL-02),” last modified Dec. 11, 2014, https://www.fda.gov/ICECI/EnforcementActions/WarningLetters/ucm434928.htm.
- U.S. Food and Drug Administration, “FDA to Investigate Added Caffeine,” last modified June 21, 2016, https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm350570.htm.
- Operation Supplement Safety, “Caffeine: What’s the Difference Between Caffeine and Caffeine Anhydrous?” last modified May 19, 2014, https://www.opss.org/node/51.
- Operation Supplement Safety, “Caffeine: What Are Some Hidden Sources of Caffeine?” last modified May 19, 2014, https://www.opss.org/node/52.
- Operation Supplement Safety, “Caffeine,” accessed May 16, 2017, https://www.opss.org/node/140.
- Alan W. Jones, “Review of Caffeine-Related Fatalities Along With Postmortem Blood Concentrations in 51 Poisoning Deaths,” Journal of Analytical Toxicology 31, no. 3 (2017): 167–72, http://dx.doi.org/10.1093/jat/bkx011.
- U.S. Food and Drug Administration, “Tragic Deaths Highlight the Dangers of Powdered Pure Caffeine,” last modified Dec. 16, 2014, https://blogs.fda.gov/fdavoice/index.php/2014/12/tragic-deaths-highlight-the-dangers-of-powdered-pure-caffeine/.
- U.S. Food and Drug Administration, “Pure Powdered Caffeine,” last modified Sept. 1, 2015, https://www.fda.gov/food/dietarysupplements/productsingredients/ucm460095.htm.
- Ibid. All five companies submitted corrective actions in response to warning letters received.
- Operation Supplement Safety, “Dietary Supplements and Drug Testing,” accessed May 16, 2017, https://www.opss.org/articles/dietary-supplements-and-drug-testing.
- Operation Supplement Safety, “Steroids: It’s Illegal to Use Anabolic Steroids, but I See Some Steroid-Like Ingredients, and Some Are Advertised as ‘Legal Steroids.’ How Do I Know if They’re Safe to Take?” accessed May 16, 2017, https://www.opss.org/faqs/steroids-its-illegal-use-anabolic-steroids-i-see-some-steroid-ingredients-and-some-are.
- 21 U.S.C. § 355.
- 21 U.S.C. § 321(ff).
- 21 U.S.C. §343(r)(6).
- 21 U.S.C. § 350b(d).
- 21 U.S.C. §§ 342 and 343.
- Josh Long, “FDA Still Finds ‘Basic’ GMP Violations in Supplement Industry,” Natural Products Insider, March 31, 2016, https://www.naturalproductsinsider.com/blogs/insider-law/2016/03/fda-still-finds-basic-gmp-violations-in-supplemen.aspx.
- Operation Supplement Safety, “Background & Overview,” accessed May 15, 2017, https://www.opss.org/about-us.