Antibiotic Prescribing Trends in U.S. Hospitals Raise Flags
Efforts needed to curb the use of broad-spectrum drugs, slow the spread of resistance
Approximately half of patients admitted to hospitals will receive antibiotics. National antibiotic use patterns illustrate the potential for individual hospitals to curb prescribing of these drugs, an essential step in slowing the spread of resistance and avoiding adverse events. An analysis from the U.S. Centers for Disease Control and Prevention (CDC) has uncovered worrisome trends in hospital prescribing; stewardship efforts should focus on reversing these trends.
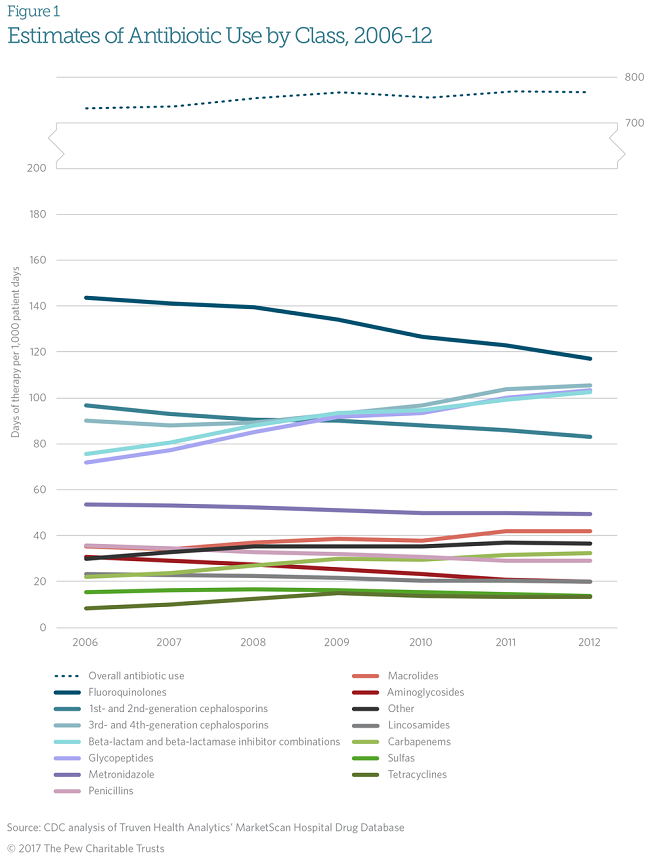
A CDC analysis of inpatient antibiotic prescribing data from the Truven Health Analytics’ MarketScan Hospital Drug Database concluded that between 2006 and 2012, overall antibiotic use remained static. (See Figure 1.) However, the prescribing of certain classes of antibiotics changed as follows:
- Use of broad-spectrum antibiotics increased significantly. Broad-spectrum antibiotics are agents that target a wide array of bacterial pathogens. They include glycopeptides, beta-lactams and beta-lactamase inhibitor combinations, carbapenems, macrolides, and third- and fourth-generation cephalosporins.
- Use of narrow-spectrum antibiotics decreased. Narrow-spectrum antibiotics are medications effective in treating a limited and targeted group of pathogens. They include penicillins, aminoglycosides, and first- and second-generation cephalosporins.
Although these estimates measure only the volume, and not the appropriateness, of prescribing, the trend toward using broad-spectrum agents is cause for concern, because these types of antibiotics have been shown to significantly increase the risk of drug-resistant infections.1
Another worrisome trend is that fluoroquinolones were the most commonly prescribed class of antibiotics in hospitals, accounting for more than 16 percent of all antibiotic use. These broad-spectrum antibiotics have significant toxicities that have been associated with serious and sometimes permanent side effects affecting tendons, muscles, joints, nerves, and the central nervous system. Because of these toxicities, the Food and Drug Administration recently issued an advisory recommending that fluoroquinolone use be avoided for patients with uncomplicated bacterial infections, such as urinary tract infections and sinusitis, and that these patients be given alternative treatment options.2 Reducing unnecessary or inappropriate use of fluoroquinolones will help minimize the risks associated with them.
New information on prescribing patterns can support hospital antibiotic stewardship programs, which are designed to optimize antibiotic use, by identifying areas to target stewardship interventions to reduce any inappropriate use.3 Further research is necessary to evaluate the appropriateness of how these drugs are used in U.S. hospitals, but the high volume of fluoroquinolone use and the increasing use of other broad-spectrum antibiotics present potential targets for antibiotic stewardship efforts.
Endnotes
- Public Health England, “Management of Infection Guidance for Primary Care for Consultation and Local Adaptation” (May 2016), accessed Nov. 16, 2016, https://www.gov.uk/government/uploads/system/uploads/attachment_data/ file/524984/ Management_of_infection_guidance_for_ primary_care_for_consultation_and_local_adaptation.pdf; Matthew E. Falagas and Petros Kopterides, “Risk Factors for the Isolation of Multi-Drug-Resistant Acinetobacter baumannii and Pseudomonas aeruginosa: A Systematic Review of the Literature,” The Journal of Hospital Infection 64, no. 1 (2006): 7–15, doi:10.1016/j.jhin.2006.04.015; Balazs Ivady et al., “Factors Influencing Antimicrobial Resistance and Outcome of Gram-Negative Bloodstream Infections in Children,” Infection 44 no. 3 (2016): 309–321, doi:10.1007/s15010-015-0857-8; and G. Gopal Rao, “Risk Factors for the Spread of Antibiotic-Resistant Bacteria,” Drugs 55, no. 3 (1998): 323–330, doi:10.2165/00003495-199855030-00001.
- Food and Drug Administration, “FDA Drug Safety Communication: FDA Updates Warnings for Oral and Injectable Fluoroquinolone Antibiotics Due to Disabling Side Effects,” accessed Sept. 15, 2016, http://www.fda.gov/Drugs/DrugSafety/ucm511530.htm; Food and Drug Administration, “FDA Drug Safety Communication: FDA Advises Restricting Fluoroquinolone Antibiotic Use for Certain Uncomplicated Infections; Warns About Disabling Side Effects That Can Occur Together,” accessed Sept. 15, 2016, http://www.fda.gov/Drugs/DrugSafety/ucm500143.htm.
- The Pew Charitable Trusts, “Antibiotic Stewardship Programs Vary in U.S. Hospitals,” 2016, http://www.pewtrusts.org/en/research-and-analysis/fact-sheets/2016/10/antibiotic-stewardship-programs-vary-in-us-hospitals.