Better Medical Device Data Yield Improved Care
The benefits of a national evaluation system
Overview
The current system for evaluating implanted medical devices provides inadequate information on the performance of the products used. This hinders prompt identification of failures and often requires lengthy and costly studies to investigate technologies both before and after approval. Better integration of medical records, registries, and other data sources would ensure that the Food and Drug Administration (FDA), manufacturers, health plans, researchers, and others can more quickly analyze device performance to improve care, accelerate patient access to innovations, and enhance safety. However, establishing this new system would require both initial funding and long-term financial support from government, private sector users, and other stakeholders.
Current device data are limited
Traditionally, manufacturers and FDA have relied on adverse event reports and clinical trials to understand the safety and effectiveness of medical devices. Those methods, however, have well-documented limitations, including that they often fail to provide robust data on large numbers of patients. On a limited basis, FDA, manufacturers, and researchers can also use various data sources—such as registries, electronic health records, and insurance claims—to evaluate the performance of medical devices. Each data source offers some insights into product performance, but in isolation is inadequate. For example, while hospital electronic health records provide rich information on patients during a particular episode of care, they often lack data from all clinicians that care for an individual after discharge. Insurance claims, on the other hand, lack detailed clinical data and do not indicate the specific device used in a procedure, but they collect information from all clinicians who care for a patient over time.
The inability to link these multiple data sources held in separate locations under the existing system makes it difficult to quickly detect safety problems and assess how well devices perform in the long term.
Existing approaches to evaluate medical devices
The currently available approaches to investigate the safety or performance of devices include the following data sources, algorithms, and tools:
- Adverse event reports. Manufacturers and hospitals must report product safety issues to FDA, but many problems go unreported or lack critical information, such as details on how the product malfunctioned, notes about the diagnosis, the procedure and medication list, and most importantly, denominator data, which indicate the total number of patients who received the device.
- Clinical studies. Device manufacturers conduct clinical trials to obtain data on the safety and effectiveness of their products before and after they go on the market. However, these trials are often small and may not be representative of the patients who will be using the device.
- Clinical data registries. Registries collect detailed clinical information on patients with similar medical conditions over a set period of time, but they are expensive to establish and operate.
- Electronic health records. Patients’ medical records contain rich data on the individual’s health. Interoperability challenges, though, hinder their ability to seamlessly exchange information with other databases and limit their usefulness when patients see multiple clinicians.
- Insurance claims. Claims data held by the Centers for Medicare & Medicaid Services and private health plans retain information across providers and over long periods of time, but this information often lacks key clinical data, including the specific brand or model of device. This makes it difficult to conduct detailed analyses.
- Electronic data aggregators. Data aggregators, such as FDA’s Sentinel Initiative or the National Patient- Centered Clinical Research Network (PCORnet), can provide insights on various health questions but cannot examine medical device performance because their data sources do not have information on the specific products used in care.
- Patient-generated data. More patients are now tracking their own health data with smartphones and mobile products than ever before. This source of real-world data has the potential to inform FDA, manufacturers, and health plans but in its current form provides little interpretable data on the performance of FDA-approved devices.
- Novel surveillance algorithms. Emerging methods to evaluate devices—such as through innovative algorithms that assess registry data and automatically alert researchers when they detect potential safety issues—can help provide information that may have otherwise been overlooked.
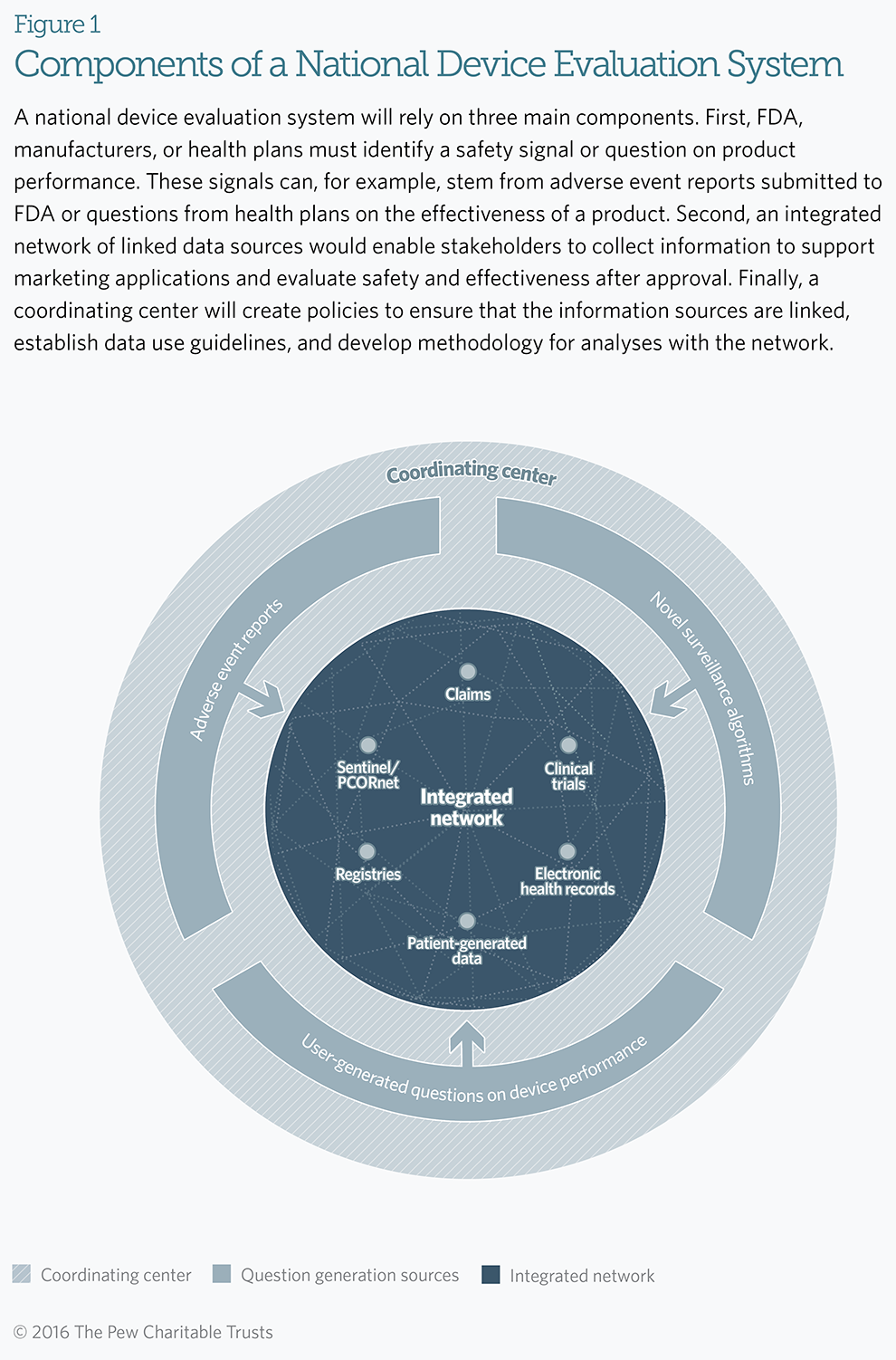
Achieving the goal of a national medical device evaluation system
Given the challenges in linking and leveraging existing information, two groups of experts convened by FDA have recommended the development of a national device evaluation system composed of three main elements.1
The first component is made up of sources that provide questions on device performance. FDA, manufacturers, health plans, and researchers first identify priorities for device evaluation and detect areas where additional research is needed. For example, adverse event reports can act as first-line identification of problems that warrant further investigation. Separately, manufacturers, health plans, and other stakeholders may have important questions that the system could answer: for instance, whether already marketed products are safe to use for indications not currently specified on the label, or how the safety of one product compares with another.
The second component answers those questions via an integrated data network. The linked network of health records, claims data, registries, and other sources can be used to evaluate device performance and safety. Through this approach, researchers at FDA, manufacturers, or health plans could pull data from multiple locations to obtain more comprehensive information on patient outcomes. Development of this integrated network would allow researchers to select the data sources they need to better understand outcomes associated with a particular product.
Lastly, the system requires a coordinating center to develop needed guidance for the use of the integrated data network. Specifically, the coordinating center must:
- Develop a sustainable funding model so that no one stakeholder bears the costs of establishing and maintaining the system.
- Create policies for the standard documentation and exchange of data, including unique device identifiers and adoption of better patient identification methods, so that information in one location is easily integrated with another source.
- Enact appropriate data use guidelines to govern how researchers can use the evaluation system while still protecting patient privacy and without burdening data holders.
- Implement methodologies to help researchers use best practices in analyzing data.
- Support a communications strategy to inform the public of important findings, including safety concerns with particular products.
The Value of a National Device Evaluation System
The following use cases illustrate how stakeholders—including manufacturers, clinicians, FDA, health plans, and researchers—could benefit from improved information on medical devices, helping patients receive the best care possible.
- Use case 1. FDA receives an increase in adverse event reports describing sudden heart failure associated with a new implanted cardiac defibrillator. The agency can then query a cardiovascular registry and Medicare claims for a detailed picture of safety over a long period of time to determine whether the device should be recalled.
- Use case 2. A private health plan seeks to evaluate whether a particular type of artificial hip fails for women with arthritis at a higher rate than implants made of different materials. The health plan supplements its own claims data, which lack clinical information, with those from several large hospitals’ electronic health record systems and pedometer data supplied by plan members to better inform coverage policies.
- Use case 3. A manufacturer of an already approved ocular implant wants to obtain an expanded indication to promote its use in pediatric patients. The manufacturer could expand its post-approval study of the product by linking with an existing ophthalmologic registry and the electronic health records of a major children’s medical center to identify pediatric patients who received the implant and examine the safety and effectiveness of this device in children.
Next steps
The creation of an integrated device evaluation system requires initial funding to establish a governance board; draft and implement a long-term business plan; develop methodologies for coding and linking information across the data sources; and institute policies, for example, on the public disclosure of data. FDA, expert groups, and others agree that resources to support this system could come from several sources, including congressional appropriations, manufacturer user fees, and individual investments from interested stakeholders that would benefit from better data on device performance and safety. With the successful launch of such a system, manufacturers would be able to more efficiently evaluate their products, and patients, health plans, and clinicians would have better assurances on the safety and performance of medical devices.
Endnote
- National Postmarket Surveillance Planning Board, “Strengthening Patient Care: Building an Effective National Medical Device Surveillance System” (February 2015), http://www.fda.gov/downloads/AboutFDA/CentersOffices/ OfficeofMedicalProductsandTobacco /CDRH/CDRHReports/UCM435112.pdf; and Medical Device Registry Task Force, “Recommendations for a National Medical Device Evaluation System: Strategically Coordinated Registry Networks to Bridge Clinical Care and Research” (August 2015), http://www.fda.gov/downloads/aboutfda/centersoffices/ officeofmedicalproductsandtobacco /cdrh/cdrhreports/ucm459368.pdf.






