Major Food Companies Committed to Reducing Antibiotic Use
Overview
Consumers across the United States are demanding meat and poultry sourced from producers who use antibiotics responsibly—and large restaurants, producers, and supermarkets are listening. Following is a list of some leading companies that have committed to using their purchasing policies to be part of the solution to the problem of antibiotic resistance. For more detail on company policies, download the pdf, which includes clickable hyperlinks.
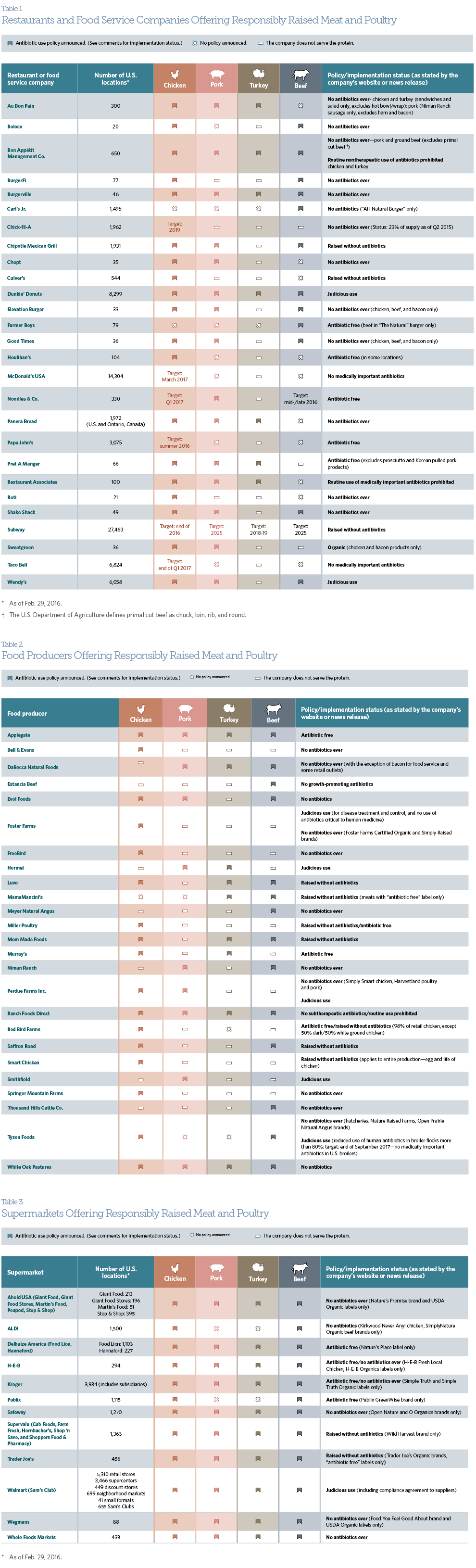
These companies’ antibiotic use policies, for major food animal species1 and associated labels, are defined by the U.S. Department of Agriculture (USDA) or the Food and Drug Administration (FDA) as follows:
- Organic:2 A standard that includes no use of antibiotics during production stages.
- No antibiotics ever/no antibiotics/raised without antibiotics:3 A label used when sufficient documentation demonstrates that no antibiotics were used during the food animal’s life.
- No medically important antibiotics:4 A label indicating that antibiotic drugs important for therapeutic use in humans cannot be used; some antibiotics with no relevance in human medicine can be used.
- Judicious use:5 A standard that follows the judicious use principles outlined by FDA and the American Veterinary Medical Association; unnecessary or inappropriate use of antibiotics should be avoided.
Other antibiotic policies and company labels may have variations of the descriptions above:6
- Antibiotic free
- Routine use prohibited
- No growth-promoting antibiotics7
Policies/offerings are subject to change. Unless otherwise noted, a company’s specifications on disease treatment, prevention, and/or control may not be articulated in its policies.
This guide is not intended to represent all companies encouraging the responsible use of antibiotics in meat and poultry production. Restaurants and food service companies listed are limited to those with at least 20 locations. The list provides examples of companies with an antibiotic policy found on their website or in a news release. Additionally, some companies provided clarification to their antibiotic policies via direct email correspondence with The Pew Charitable Trusts prior to Feb. 29, 2016. To add your company’s name to this list, please contact [email protected].
Endnotes
- Defining “Small Number of Animals” for Minor Use Designation, 74 Fed. Reg. 43043 (Aug. 26, 2009), https://www.gpo.gov/fdsys/pkg/FR-2009-08-26/html/E9-20553.htm.
- U.S. Department of Agriculture, “Organic Livestock Requirements” (July 2013), https://www.ams.usda.gov/sites/default/files/media/Organic Livestock Requirements.pdf.
- No antibiotics ever/no antibiotics/raised without antibiotics are company-defined labels that follow that company’s standards on antibiotic use. The labels may be independently verified through the USDA Process Verified Program. U.S. Department of Agriculture, “Meat and Poultry Labeling Terms,” last modified Aug. 10, 2015, http://www.fsis.usda.gov/wps/portal/fsis/topics/food-safety-education/ get-answers/food-safety-fact-sheets/food-labeling/meat-and-poultry-labeling-terms/meat-and-poultry-labeling-terms.
- U.S. Food and Drug Administration, “Guidance for Industry #209—The Judicious Use of Medically Important Antimicrobial Drugs in Food-Producing Animals,” last modified April 13, 2012, http://www. fda.gov/downloads/AnimalVeterinary/GuidanceComplianceEnforcement/ GuidanceforIndustry/UCM216936.pdf.
- Ibid. and U.S. Food and Drug Administration, “Guidance for Industry #213—New Animal Drugs and New Animal Drug Combination Products Administered in or on Medicated Feed or Drinking Water of Food-Producing Animals: Recommendations for Drug Sponsors for Voluntarily Aligning Product Use Conditions With GFI #209,” last modified December 2013, http://www.fda.gov/downloads/ AnimalVeterinary/GuidanceComplianceEnforcement/GuidanceforIndustry/ UCM299624.pdf.
- The labels listed are claims that adhere to company antibiotic policies but are not approved and/or verified by the USDA.
- FDA has offered guidance to facilitate the phasing out of medically important antibiotics for production purposes in “Guidance for Industry #213,” a follow-up to “Guidance for Industry #209” that will take effect Jan. 1, 2017.









