Charges Made for Deadly Outbreak Linked to Compounded Medicine
Fourteen people—including former pharmacists, pharmacy technicians, and the owners of the New England Compounding Center in Framingham, Massachusetts—were charged Dec. 17 for their alleged roles in a 2012 outbreak of spinal meningitis that sickened more than 750 people and killed 64. Contamination of compounded steroid injections led to a recall of all the company’s products in fall 2012.
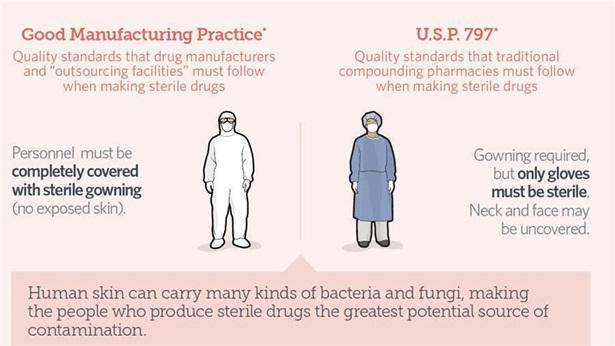
The meningitis outbreak called into question the efficacy of compounding oversight. Pharmacy compounding operations have expanded significantly in recent decades, resulting in some production facilities that operate more like pharmaceutical manufacturers in scale but lack the same supervision or quality standards.
"When you're compounding on that different scale [and] you're reaching more patients, you really do need to be meeting higher quality standards," Gabrielle Cosel, who manages Pew’s drug safety project, told Modern Healthcare.
Since 2001, compounding errors have been associated with at least 1,049 adverse events, including 89 deaths. The Drug Quality and Security Act, signed into law by President Barack Obama in 2013, was designed to improve the safety of the U.S. drug supply and contains important updates to the oversight systems for pharmaceutical compounding.
The law established a new regulatory category for pharmaceutical compounders that supply sterile medicines for use in hospitals, doctors' offices, and clinics. These “outsourcing facilities” will meet the same standards as pharmaceutical manufacturers, providing a safer source of drugs compounded at larger scales.