Conflicts-of-Interest Policies for Academic Medical Centers
Recommendations for Best Practices
Principled scientific collaboration between physicians and the drug and medical device industries has contributed to many of the medical advances of the past century—penicillin and implantable cardiac pacemakers, to name just two. Industry marketing efforts, however, have increasingly slipped into the educational and clinical programs of academic medical centers.
Webinar: Protecting the Integrity of Medical Education
 © 2009 National Geographic
© 2009 National GeographicIn addition, pressure from the U.S. Office of the Inspector General,1 congressional hearings,2 and lawsuits alleging deceptive marketing practices3 led the Pharmaceutical Research and Manufacturers of America to recommend that member companies cease providing noneducational gifts and certain types of free meals to physicians.4
In 2012, The Pew Charitable Trusts convened an expert task force on conflicts of interest in medicine to assist academic medical centers in identifying best practices to incorporate into their conflict-of-interest policies. The panel reviewed the literature and consulted experts in order to create an updated list of recommendations to those established by the Association of American Medical Colleges in 2008. The task force formulated recommendations in 15 areas in order to protect the integrity of education and training and the practice of medicine within the academic medical center while not standing as an impediment to research and scientific inquiry.
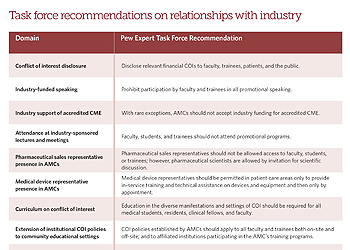
The best-practice recommendations are:
- Acceptance of gifts and meals: No gifts or meals of any value should be accepted by clinical faculty members and staff, medical students, residents, clinical fellows, or other clinical trainees from the pharmaceutical, biotechnology, medical device, or medical diagnostics industries or their sales representatives.
- Disclosing conflicts of interest: Faculty should be required to disclose to their institutions all industry relationships that relate to their academic activities in teaching, research, patient care, and institutional service.
- Industry-funded speaking: Faculty should not accept industry funding for speaking engagements directed toward other faculty, medical students, trainees, patients, community physicians, health professionals, or the public.
- Continuing medical education: In general, continuing medical education should not be supported by industry.
- Attendance at industry-sponsored lectures and meetings: Faculty, students, and trainees should not attend promotional or educational events that are supported directly by industry.
- Pharmaceutical sales representative presence in academic medical centers: Pharmaceutical sales representatives should not be allowed access to any faculty, students, or trainees in academic medical centers or affiliated entities.
- Medical device representative presence in academic medical centers: The access of medical device representatives to patient care areas should be limited to in-service training and technical assistance on devices and other equipment already purchased and then only by appointment and with disclosure to and consent from the patients who would be involved.
- Curriculum on conflict of interest: Conflict-of-interest education should be required for all medical students, residents, clinical fellows, and teaching faculty.
- Extension of institutional conflict-of-interest policies to community educational settings: Conflict-of- interest policies established by academic medical centers should apply to all faculty members regardless of the nature of their relationship to the institution—paid or voluntary, full time or part time, on-site or off-site— and to affiliated institutions participating in the academic medical center's educational and training programs.
- Industry-supported clinical fellowships: In general, clinical fellows, residents, and medical students may not accept industry-sponsored fellowships earmarked specifically for clinical training but may compete for industry fellowships awarded for scientific training.
- Ghostwriting and honorary authorship: Academic medical faculty and trainees should follow the International Committee of Medical Journal Editors standards for authorship and contributorship. Ghostwriting and honorary authorship are strictly prohibited.
- Consulting relationships for research and scientific activities: Faculty and trainees should be permitted to engage in consulting relationships with pharmaceutical and device companies about research and scientific matters.
- Consulting relationships for marketing (excluding scientific research and speaking): Academic medical faculty and trainees should be prohibited from engaging in consulting relationships that are solely or primarily for commercial marketing purposes.
- Pharmaceutical samples: An academic medical center should not accept samples unless it determines that there are compelling circumstances to do so. In these cases, it should implement mechanisms for accepting samples that prevent their use as marketing tools.
- Pharmacy and therapeutics committee: Ideally, voting members of these committees should not have a financial relationship with industry. In circumstances when this standard cannot be achieved, members with such relationships should be recused from any discussion of, or voting on, a related product, whether the product is manufactured by the company, is a competitor of that product, or is in the same class as that product. All committee members should disclose financial relationships with pharmaceutical and medical device companies, as should practitioners requesting changes or additions to the institution's formulary.
This report will discuss these best-practice recommendations and provide the reasoning behind their formation. Included will be case studies at academic medical centers and examples of conflict-of-interest policies that are currently in place.
* The term “conflict-of-interest policies” refers to policies regulating the financial relationships between health care companies and academic medical center staff and trainees in the areas of medical education and clinical care. These are also known as “clinical conflicts of interest.” Policies relating to conflicts of interest in medical research are not addressed in this report.
A shorter version of these best practice recommendations can be found in the December 2013 issue of the Journal of the American Medical Association.
Many medical schools and medical centers have adopted conflict-of-interest policies banning various industry inducements to influence students, trainees, and faculty clinical practice. The Pharmaceutical Research and Manufacturers of America, the trade association for drug companies, has also recommended that its member drug companies stop providing noneducational gifts and certain types of free meals to physicians.
It remains unclear, however, how widespread and effective these changes have been. The Institute on Medicine as a Profession, which maintains a comprehensive database of medical school conflict-of-interest policies,5 reported that the percentage of medical schools with strict policies increased from 10 percent in 2008 to 27 percent in 2011 but that there are still areas that need to be addressed, such as ghostwriting; free samples; continuing medical education, or CME; consulting; honoraria; and speakers' bureaus.6
In 2008, the American Medical Student Association, or AMSA, with support from The Pew Charitable Trusts, created a medical school “scorecard” that annually assigns a letter grade to institutions based on how fully they comply with the Association of American Medical Colleges' recommendations, which included restrictions or prohibitions on a number of practices, such as industry gifts and meals, speakers' bureaus, and the presence of pharmaceutical sales reps on campus.7 According to the scorecard, the percentage of medical schools that have adopted policies consistent with the Association of American Medical Colleges' recommendations, corresponding to an A grade, increased from 4.7 percent in 2009 to 25.3 percent in 2012.8
The policy provisions with the most dramatic improvements in grades have been those banning outright payments and gifts from industry. Conversely, relatively few schools have implemented policies restricting promotional speaking, eliminating industry support of accredited continuing medical education, and banning access to the institution by pharmaceutical sales representatives. In addition, even when schools have adopted well-crafted policies, compliance is not assured. One recent survey of medical schools, for example, found that up to half of medical students and residents reported receiving gifts from pharmaceutical companies, even in schools graded highly on the AMSA scorecard.9 Thus, while institutions can set guidelines and provide a level of oversight and enforcement, it is not clear that these are sufficient to change an individual's behavior, much of which can occur outside of the institution's purview.
All policies were decided by consensus. The format of each section includes the final best-practice recommendation and a brief discussion of the rationale for the policy decision. In most cases, the section includes an example of a policy in place at an academic medical center. These are not intended as “model” policies, but rather are case studies of how multiple institutions are addressing the nuances of conflict-of-interest policies, sometimes in a manner that is influenced by their local environments and issues.
1. Acceptance of gifts and meals
No gifts or meals of any value should be accepted by academic medical center clinical faculty members and staff, medical students, residents, clinical fellows, or other clinical trainees from the pharmaceutical, biotechnology, medical device, or medical diagnostics industries or their sales representatives. This prohibition includes gifts that may be deemed “educational” for medical staff, such as textbooks, but does not prohibit patient educational material.
Gifts and meals of any size from the pharmaceutical industry have been shown under some circumstances to be associated with positive attitudes toward sales representatives, the addition of promoted drugs to hospital formularies, preferential prescribing of brand medications rather than available generics, and nonrational prescribing practices such as not following national guidelines when choosing which medications to use.10 A more general body of social science research indicates that gifts influence individuals to respond reciprocally, by returning the favor.11
Relationships between industry marketing and academic medical centers peaked in the mid-2000s when studies revealed that 96.8 percent of third-year medical students reported receiving industry-sponsored free lunches,12 40 percent of emergency medicine residencies allowed industry to fund social events for residents,13 88 percent of psychiatric residencies accepted 4.8 industry-sponsored lunches per week,14 and 48 percent of all income for accredited continuing medical education came from industry sources.15 Policy bans of gifts and free meals have now become common in medical schools, with more than 60 percent (93 of 153 medical schools) adopting such policies, according to the 2013 American Medical Student Association scorecard16—a positive result but with room for improvement.
One point of continuing controversy is the appropriateness of free “modest meals” as part of industry-funded Accreditation Council for Continuing Medical Education-sanctioned courses. Most institutions do not include these meals in their general bans on industry-funded gifts, perhaps because the industry money for the meals is often channeled through a third party such as a medical education and communication company. However, because the council's standards for commercial support require that all learners be informed in advance of the financial source of a course, attendees know who is funding the meal. Therefore, to minimize any sense of reciprocity, the task force recommends that physicians be required to pay for their own meals at such events.
An exception to this prohibition should be industry-purchased meals as part of contracts for consultation or research, in which food is part of fair market compensation for clearly delineated services and not a gift.
Example policy: Penn State University College of Medicine/ Penn State Milton S. Hershey Medical Center*
“Gifts from industry are a form of marketing intended to create a relationship that can influence
decision-making. Though it is hard to know how much any particular decision is influenced by receipt of a gift, the practice has been shown to influence physicians' behavior in the aggregate.
“A. As such, industry representatives are prohibited from giving any form of gift to physicians and other faculty, staff, students, and trainees on the PSHMC campus.
“B. Physicians and other faculty, staff, students, and trainees may not accept any gifts from industry representatives on the PSHMC campus, including meals and gifts of trivial material value.
“C. Moving an interaction with industry representatives to an off-campus location in order to avoid these restrictions would be in violation of this policy.
“D. When attending off campus meetings, conferences, etc., faculty, staff, students, and trainees are discouraged from accepting gifts (which includes meals) from industry representatives.
“E. Gifts containing industry brands and/or logos such as clocks, posters, post-it pads, etc. are marketing tools, and should not be displayed in clinical and teaching areas. Anatomic models or charts that are deemed critical for patient education are permitted, but non-branded versions are preferred.
“F. Employees and trainees may not accept any form of compensation for listening to a promotional presentation (‘detailing') by an industry representative.
“G. Employees and trainees may not accept any form of compensation or rebate for prescribing or changing a patient's prescription.
“H. Even if a gift is covered by an exception, staff may not accept a gift if it would undermine the appearance of PSHMC's integrity.
“II. Food
“A. Industry-sponsored meals in the workplace are not permissible on campus, with the exception of food provided in connection with Accreditation Council for Continuing Medical Education (ACCME) accredited programming and in compliance with ACCME guidelines.”
* Industry Relations Policy, Penn State Milton S. Hershey Medical Center, accessed Nov. 8, 2013, http://www.pennstatehershey.org/c/document_library/get_file?uuid=dd1ad7cc-8dea-4b8f-8d67-3ca3c727a172&groupId=867743.
2. Disclosing conflicts of interest
The faculty at academic medical centers should be required to disclose to their institutions all industry relationships that relate to their academic activities in teaching, research, patient care, and institutional service and to inform students and trainees of the nature of those interests, including specific companies and therapeutic products and how they relate to the educational topics, whether the session is a lecture, seminar, rounds, or other format. When a financial interest relates to potential patient treatments, faculty should inform patients of these interests in a similar way, where appropriate and feasible.
The task force encourages public disclosure of financial relationships on a public website, but acknowledges that institutions may choose other methods of disclosure.
Disclosure is an important component of a conflict-of-interest policy. Faculty should be transparent about any financial interests that could potentially influence their professional judgments and choices. Disclosure of financial relationships has long been required in various spheres of medical activity, including when submitting journal articles, applying for research grants, and participating in accredited continuing medical education courses. All academic medical centers that receive National Institutes of Health funding must require some form of annual disclosure to the institution of faculty members' financial interests that relate to their academic duties. Typically these disclosures are kept confidential and are referred to by compliance officers when the faculty apply for or become engaged in federally funded research.
While all institutions that apply for federal (and increasingly, nonfederal) funding for research require that faculty members at least annually disclose relevant financial interests to internal compliance officials, external disclosure is less common. There are three types of external disclosure that institutions should require: disclosure to staff, students, and trainees; disclosure to patients and research subjects; and disclosure to the public at the time of any presentation of the research, written or verbal.
Increasingly, students are requesting that faculty disclose financial relationships before preclinical and clinical teaching activities.17 The effect of direct disclosure to medical students, for example, was studied in a recent comparison of student attitudes before and after a stringent disclosure policy was instituted at Mount Sinai School of Medicine in 2010. The school's policy mandates that lecturers disclose their related financial conflicts of interest to preclinical medical students before lectures. Nearly all the medical students (97 percent) favored such a disclosure policy, and after it was implemented, students were more likely to support policies banning interactions of both faculty and students with drug or device sales representatives. They were not more likely, however, to disapprove of general interactions between faculty and industry.18
Disclosure to trainees can occur in the setting of didactic lectures, and if so, we recommend that the information include the name of the company, the nature of the relationship, the name of the associated drug or device, and the approximate dollar value of the relationship, where relevant or pertinent—that is, when the subject of the lecture is related to the use of the drug or device. Such disclosure is also important during the more informal setting of clinical rounds.
Disclosure to patients is uncommon but may occur in certain situations. For example, the American Academy of Orthopaedic Surgeons advises in its code of ethics that orthopedists disclose directly to their patients all financial relationships with vendors. Such disclosure can be in the form of a handout given directly to patients as they register for appointments or posted in a prominent place in the waiting room. More research is needed to understand the best modes and content of such disclosures. Communication from providers to patients should support the patients' independent evaluation of the potential impact of financial relationships on their care.
Finally, some institutions post faculty financial interests on a public website. The Physician Payments Sunshine Act mandates that all companies that participate in U.S. federal health care programs disclose payments to physicians; these transparency reports will be posted on the federal Open Payments website as of September 2014. Some institutions may choose to avoid the expense of operating their own disclosure websites and wait for the federal site to become functional. However, there are disadvantages to the Open Payments reports. For example, all data on the website will be six to 18 months out of date because transparency reports reflecting a year's payments will not be posted until June 30 of the subsequent year. In addition, the Physician Payments Sunshine Act does not require disclosure of industry payments to nonphysician health care professionals, such as nurses, pharmacists, and physician assistants.
Example policy: Johns Hopkins Medicine*
“Faculty must disclose all relevant payments from and interests in industry in related presentations and publications and in lectures to students and trainees.”
* Johns Hopkins Medicine Policy on Interaction With Industry, Johns Hopkins Medicine, accessed Nov. 8, 2013, http://www.hopkinsmedicine.org/Research/OPC/Policy_Industry_Interaction/policy_interaction_industry.html.
3. Industry-funded speaking
Faculty should not accept industry funding for speaking engagements directed toward other faculty, medical students, trainees, patients, community physicians, health professionals, or the public. This restriction should include both formal speakers' bureaus and all other speaking arrangements in which the company has any influence or control over the content. This restriction would not apply to formal industry research-and- development contracts, nor would it include scientific presentations made to industry employees.
Industry-funded speaking, as opposed to industry-supported accredited continuing medical education courses, refers to arrangements, usually through speakers' bureaus, in which a company directly pays a physician to speak. Arrangements with speakers' bureaus typically include three components:
-
A financial contract exists between a company and the speaker.
-
The content of the talk is regulated by the U.S. Food and Drug Administration, or FDA, as a promotional speech under the Food, Drug, and Cosmetics Act, which means it cannot contain discussion of off-label, or nonapproved, uses of drugs or devices.19
-
The speaker must allow the company to review the presentation's content in advance, and in some cases, as is the policy at GlaxoSmithKline, the company requires that the speaker use company-prepared slides.20
Faculty who participate in speakers' bureaus are de facto “marketers in academic robes” and lend a patina of academic endorsement to the promotional agenda of the sponsoring companies, which compromises academic integrity. Furthermore, promotional speakers are poor role models for trainees. By using text and slides provided by the sponsoring company, they may fail to conduct an independent appraisal of the medical evidence, undermining a fundamental teaching obligation of medical schools.
Among those academic medical centers that have sought to discourage participation in speakers' bureaus, there is significant variation in how these speaker arrangements are described, which can lead to confusion and disagreements regarding what constitutes a banned activity. A policy that prohibits faculty from accepting industry payment for any presentations to health care practitioners would most reliably prevent promotional speaking engagements. For those academic medical centers that allow for select industry-funded talks, the policies should specifically prohibit faculty participation in any activity that is regulated by the FDA as a promotional program or for which the content is reviewed for approval or provided by the sponsoring company.
For those cases in which faculty discuss their own research, use their own slides, and assure regulatory staff that all opinions expressed are strictly their own, payment by industry would be permissible if it is part of a research or scientific consulting contract. However, direct payments for speaking outside the bounds of such a consulting or research contract is typically regulated by the FDA as promotional activities and would not be permitted under an optimal conflict-of-interest policy.
Example policy: Temple University School of Medicine*
“A. Faculty may not give compensated presentations or accept honorarium, food, lodging or transportation for participation in non-CME events directed at physicians or other health care professionals, such as restaurant talks. It is acknowledged that consultation for industry may require presentations.
“B. Faculty may not accept compensation for membership on a speakers list (speakers' bureau), nor accept any compensation from industry for attending any industry-sponsored event at which he/she is not a speaker. This includes industry support to defray costs of attending such meetings, e.g., travel, hotel, and meals.”
* Temple University School of Medicine Procedures Implementing the Temple University Policy and Procedures on Conflict of Commitment and Conflict of Interest (Policy Statements A-H) Including Financial Conflicts of Interest Related to Research (Policy Statement I), Temple University School of Medicine, accessed Nov. 8, 2013, http://www.temple.edu/medicine/faculty/documents/conflict_policy.pdf.
4. Continuing medical education
In general, continuing medical education should not be supported by industry. This would include lectures or online discussions of the medical literature that make recommendations regarding which drugs or medical devices are most appropriate for various patients. In situations where industry funding is nonetheless being considered, academic medical centers should implement additional safeguards beyond compliance with the Accreditation Council for Continuing Medical Education's Standards for Commercial Support. Examples of such safeguards might include: creating a “blinded” pool of industry funds; requiring that any activity be funded by more than one company; calling for physicians to use some of their own money (such as paying for their own meals); and locating the continuing medical education activity within an academic setting or other appropriate venue conducive to education.
Some types of technical training, such as training in devices, might require support from industry to defray expenses and provide technical expertise. Academic medical centers should judge the necessity of accepting industry funding for such training programs on a case-by-case basis, taking into account the necessary expenses and whether industry involvement is required for providing adequate hands-on training. Any industry support accepted should be limited to funding of educational programming and not amenities, and such courses should be audited carefully by academic medical centers for commercial bias.
Accredited continuing medical education receives substantial financial support from industry. In 2007, for example, industry educational grants provided 46 percent of all CME funding; this proportion has gradually declined to 27 percent in 2012.21
ACCME Standards for Commercial Support were promulgated to prevent industry's promotional content from entering accredited continuing medical education. Key requirements include financial disclosure, confirmation that supporting companies have no influence over the content and choice of speakers, and a procedure for resolving conflicts of interest should they arise.
However, studies suggest that industry funding of continuing medical education tends to bias topic choices and content in favor of the sponsors' products and therapeutic areas. Of the three published studies that have examined whether industry sponsorship is associated with higher prescribing of the sponsors' products, all three have shown a positive correlation, with higher prescribing of the sponsors' antihypertensives,22 antibiotics,23 and antipsychotics.24 Another study found that industry support of primary care continuing medical education resulted in a narrow list of course topics related exclusively to products marketed by the sponsors.25 A 2008 literature review commissioned by ACCME reviewed these studies and concluded that industry-supported continuing medical education activities were indeed effective in increasing attendees' prescriptions of the sponsors' products.26
While the empirical research on bias in industry CME is limited and in some cases outdated, the existing evidence recently led the American Medical Association to adopt ethical guidelines discouraging both industry funding of these programs and the participation of lecturers who have industry relationships.27
The task force recommends that, in general, academic medical centers should ban commercial funding of continuing medical education. However, in some situations, such as training in the use of certain medical devices, industry funding and technical expertise may be needed.
To supplement ACCME standards, institutions that find it necessary to continue to accept industry support for continuing medical education should consider mechanisms to minimize commercial bias, including the following:
-
Create an undesignated, or blinded, pool of money contributed by multiple companies for the purpose of supporting an institution's CME program. This is different from the common arrangement in which an academic medical center creates a central continuing medical education office to accept and administer commercial support of programs. Such central offices do not prevent companies from specifying the topic of the course or which academic department should receive the funds. A truly blind pool is a “no strings attached” donation of money for continuing medical education, and these funds are less likely to be allocated in ways that are biased in favor of the company's products. Three institutions that have created such pools are Stanford University (http://med.stanford.edu/news/all-news/2010/09/stanford-geneticist-wins-macarthur-genius-grant.html), Partners HealthCare (http://www.partners.org/Assets/Documents/About-Us/OII/OII_Policy.pdf), and Kaiser Permanente, a large integrated health care delivery system. It is unclear whether companies are likely to contribute to such funding pools when a more prudent investment may be for courses focused specifically on a disease area for which they are marketing a product.
-
Require that commercially funded courses be supported by at least two companies and that no single company contribute more than 50 percent for a given program. While such arrangements may minimize bias in favor of a single company's product, it may still result in bias in favor of a particular class of products, or, more fundamentally, it may produce a bias in favor of pharmacologic- or device-based treatment over lifestyle-based preventive treatments.
-
Require that physicians personally contribute some money toward industry-supported courses. Both social science and neuroimaging data provide evidence that people have a deeply ingrained sense of reciprocity. Individuals are inclined to respond to an act of kindness or a gift by returning the favor.28 Requiring physicians to pay even a small fee for a service such as a continuing medical education course might reduce the sense of reciprocal obligation. Some pharmaceutical firms have changed their educational grant policies to no longer allow funding of food and beverages for course attendees.29
-
Require that industry-supported courses take place in non-resort locations. This requirement serves to minimize the impression that one has received a gift such as a vacation from the company. In addition, this policy ensures that the primary goal is the educational experience rather than a vacation with some educational content.
Case study: University of Michigan*
In the summer of 2010, the University of Michigan Medical School announced that the university would no longer accept industry support for continuing medical education activities beginning in January of 2011.30 This decision was informed by the release of the Association of American Medical Colleges' recommendations that medical schools improve their oversight of continuing medical education courses as well as the core philosophy among academic leadership that objective medical education is a key mission of the institution.
While department chairs voted unanimously in favor of the policy, many faculty members pointed out that their external continuing medical education programs attracted physicians in the community who became sources of patient referrals for clinical and research programs at the university. To address this concern, the university created a grant program with $750,000 in initial funding split equally among the dean's office, the marketing department, and the hospital. Faculty were encouraged to use the grants to fund continuing medical education courses and demonstrate that their programs did, indeed, lead to increased referrals. Two years after the policy went into effect, no faculty could demonstrate that the programs generated more business for the school. The total number of programs was minimally affected since most are grand rounds programs held at the university that were never dependent on industry support. A small proportion of programs were external courses often held at resorts; the number of these courses decreased from approximately 60 per year to 30 per year.
* Terry Kowalenko, M.D., director of continuous professional development, University of Michigan School of Medicine, personal communication to Dr. Daniel Carlat, Jan. 31, 2013.
Example policy: University of Michigan Medical School*
“Effective January 1, 2011, UMMS-offered activities for which CME credit is designated may not receive financial funding (e.g., grant, gift, subsidy, or exhibit fee) from commercial entities that produce, market, re-sell, or distribute health care goods or services consumed by, or used on, patients or biomedical research subjects.”
* University of Michigan Medical School Policy on Industry Funding for UMMS-Offered CME, University of Michigan Medical School, accessed Nov. 8, 2013, http://www.amsascorecard.org/documents/0000/0634/CME_Policy.pdf.
While there has been no systematic survey assessing which academic medical centers receive commercial support for continuing medical education, some institutions have effectively ended such support. They include Memorial Sloan-Kettering Cancer Center (2008), Stanford University Medical Center (2008), University of Michigan Medical School (2010), Kaiser Permanente, and Brody School of Medicine of East Carolina University, which has an informal practice (although not formalized as an institutional policy) of accepting no commercial support for its programs.31 In addition, Stanford University Medical Center and Kaiser Permanente, a large integrated health care delivery system, have limited all commercial funding to a blinded central pool over which companies have no control. Of these two institutions, only Stanford has thus far received a contribution to this fund.
5. Attendance at industry-sponsored lectures and meetings
Faculty, students, and trainees should not attend promotional or educational events that are supported directly by industry. An exception is attendance at academic events, such as grand rounds and professional society meetings, in which a third party may be funded by industry to provide medical education, whether CME- accredited or not.
Just as it is inappropriate for academic medical center faculty to speak at industry-funded marketing or educational events, it is also inappropriate for faculty or trainees to attend such events. The reasons for the prohibitions are identical: FDA regulations require that industry-funded talks focus on approved uses of the products marketed by the sponsoring company, which ensures that the education provided is narrow.32 The purpose of medical education, however, is to instruct broadly on the optimal care of patients, which typically entails considering and comparing many treatment options beyond that offered by a single company.
Example policy: Stony Brook University*
“Faculty, residents, and students are discouraged from attending industry sponsored dinners or social events off campus unless these events meet the standards for accredited CME activities.”
* Stony Brook University Hospital Graduate Medical Education Policies and Procedures, Stony Brook University Hospital, accessed Nov. 8, 2013, http://medicine.stonybrookmedicine.edu/system/files/Pharmaceutical%20Vendor%20Relationships09.pdf.
6. Pharmaceutical sales representative presence in academic medical centers
Pharmaceutical sales representatives should not be allowed access to any faculty, students, or trainees in academic medical centers or affiliated entities. However, faculty may invite pharmaceutical scientists for specific educational or scientific discussions that do not involve marketing of a specific product.
Traditionally, physicians have relied heavily on meetings with pharmaceutical sales representatives for information on new drug treatments.33 However, research indicates that interactions with sales reps detract from the rational practice of medicine. A systematic review analyzed 29 studies that measured physician knowledge or prescribing patterns after such interactions and found that, in general, physician exposure to sales reps led to the prescribing of lower quality, higher frequency, and higher cost therapeutics compared with nonexposed physicians.34 Of even greater concern is evidence that sales reps may intentionally omit information35 or provide misinformation36 to increase prescriptions for their products.
While sales reps provide current information on their company's products—new dosage forms, newly described side effects, and new indications—trainees at academic medical centers can obtain such information in more efficient and objective ways, such as by Web-based resources created without industry funding.37
Faculty may appropriately choose to meet with pharmaceutical-industry scientists for purposes such as discussing potential research collaborations or receiving in-depth educational information about the company's products and therapeutic areas. Such meetings may serve important research and educational functions and should be allowed as long as they are at the invitation of the faculty member and do not include sales reps.
Some academic medical centers that find it infeasible to exclude sales reps because of internal resistance may adopt second-tier policies to control access. In such situations, acceptable policies include:
Restrict interactions. The presence of sales reps should be limited to minimize their influence on medical education. They should be permitted in the health center by appointment only and required to sign in at a designated office and wear an identification badge that clearly identifies them as vendors. Sales reps should never be permitted in patient care or designated medical education areas.
Supervise trainees. If trainee interaction with vendors is allowed to occur, it should be viewed as an educational experience and monitored by faculty. Supervision should focus on helping the trainee judge the veracity and completeness of the information received, evaluating alternatives including generic drugs and nondrug therapies, and understanding the comparative risks, costs, and benefits.
Example policy: The Commonwealth Medical College*
“Site Access for Industry Representatives:
“Pharmaceutical and device representatives are not allowed to meet with faculty regardless of location, or are not permitted to market their products anywhere inside the medical center and associated clinics and offices. (Exceptions may be made for non- marketing purposes, such as training on devices or equipment.) All Industry professionals wishing to gain access to TCMC designated sites will be required to check into the facility through a centralized, appointed individual. Purposes which are appropriate for site visits include the exchange of scientific information, dissemination of materials/information regarding new therapeutic options, and training or discussions which can lead to the advancement of healthcare. Name badges are required for all industry personnel when visiting a TCMC site. Industry representatives are prohibited from roaming areas frequented by clinicians or faculty. They may provide informational material, such as product literature or journal articles, only at the request of a faculty or staff member. Industry representatives are not permitted to market their products anywhere inside the medical sciences building and associated clinics and offices, with the exceptions of the above listed activities. Prior to gaining access, the individual must have a scheduled appointment with appropriate TCMC personnel. There may be designated times for Industry representatives to convene in a specific location as pre-determined by department heads in order for questions to be answered or for information to be disseminated regarding new equipment or therapeutic options. Any marketing activities will be limited as per sections 1 and 3 of this policy.
“Upon an initial visit to a TCMC site, industry representatives will be provided a distributor policy sheet which will outline procedures that they must follow while visiting the facility.”
* Policy on Conflicts of Interest and Interactions between Industry and Faculty, Staff and Students of the Commonwealth Medical College, The Commonwealth Medical College, accessed December 6, 2013, http://thecommonwealthmedical.com/oth/Page.asp?PageID=OTH000456.
7. Medical device representative presence in academic medical centers
The access of medical device representatives to patient care areas should be limited to in-service training and technical assistance on devices and other equipment already purchased, and then only by appointment and with disclosure to and consent from the patients who would be involved.
While medical device representatives are generally salespeople, they differ from pharmaceutical sales representatives in that they are more likely to provide valuable technical assistance that cannot easily be obtained from other sources. Therefore, policies addressing site access for medical device representatives should acknowledge that there is a legitimate relationship that requires flexibility around access to ensure optimal patient care.
Case study: Beth Israel Deaconess Medical Center*
At Beth Israel Deaconess Medical Center, medical device representatives, often termed “industry employed allied professionals,” are part of a vital yet controlled partnership with cardiologists who specialize in electrophysiology—the treatment of heart rhythm problems using implantable devices. After a physician chooses the brand and model of a device implant, a representative of the device manufacturer will attend the procedure. These industry professionals provide important technical expertise during the procedure, register patients' devices with the manufacturer, and assist with device programming. Their presence is valuable to successful outcomes because the features of devices are frequently modified and updated, as is the case with the programming options and battery properties of pacemakers and implantable defibrillators. In addition, the real-time access to specialized expertise that they provide at Beth Israel is essential to providing high-quality clinical care.
* Daniel B. Kramer, M.D., Cardiovascular Institute, Beth Israel Deaconess Hospital, personal communication to Dr. Daniel Carlat, 2013.
Example policy: Stanford School of Medicine*
Stanford's policy pertaining to medical device representatives covers industry's interactions with the School of Medicine, Stanford Hospital and Clinics, and Lucile Packard Children's Hospital. Site access is allowed, with an appointment, only for in-service device trainings and the evaluation of new purchases. The policy is as follows:
“Site Access by Sales and Marketing Representatives
“A. Sales and marketing representatives are not permitted in any patient care areas except to provide in- service training on devices and other equipment and then only by appointment.
“B. Sales and marketing representatives are permitted in non-patient care areas by appointment only.
Appointments will normally be made for such purposes as:
“1. In-service training of Stanford Hospital and Clinic or Lucile Packard Children's Hospital personnel for research or clinical equipment or devices already purchased.
“2. Evaluation of new purchases of equipment, devices, or related items.
“3. Appointments for these purposes may be made on a per visit basis or as a standing appointment for a specified period of time, at the discretion of the faculty member, his or her division or department, or designated hospital personnel issuing the invitation and with the approval of appropriate hospital management.”
* Stanford School of Medicine Industry Interactions Policy-Conflicts of Interest Disclosure, Stanford School of Medicine, accessed
Nov, 8, 2013, http://med.stanford.edu/coi/siip/policy.html#i.
8. Curriculum on conflict of interest
Conflict-of-interest education should be required for all medical students, residents, clinical fellows, and teaching faculty.
Understanding interactions between industry and health care practitioners should be a part of professional training and conveyed informally by role modeling and formally via a defined curriculum. A formal curriculum on conflict of interest aims to teach trainees and faculty how to prevent marketing activities from inappropriately influencing their treatment decisions. In general, studies on the effectiveness of such courses have shown mixed results, with some educational interventions producing more skepticism about industry marketing, some showing no change, and others resulting in a more favorable view toward industry.38 Curricula that included participation by industry employees tended to produce the most favorable attitudes toward industry; however, variability in educational interventions makes it difficult to provide clear recommendations. While empirical studies endorsing specific educational techniques are scant, some type of conflict-of-interest training in regard to research, education, and clinical practice should be offered in all academic medical centers. Training on conflict of interest in research is already required of all researchers funded by the National Institutes of Health and other U.S. Department of Health and Human Services agencies.39
While nearly half of medical schools report having some type of conflict-of-interest curriculum,40 the content varies widely. In many cases, the instruction consists only of a review of the institution's policies. Other schools have developed robust didactic courses on pharmaceutical marketing techniques and have sometimes included proctored experiences in which trainees interact either with pharmaceutical sales representatives or with faculty playing the part.
Case studies: Albert Einstein College of Medicine, the University of Iowa Carver College of Medicine, and the Geisel School of Medicine at Dartmouth University*
Schools are teaching courses on conflict of interest, ranging from the first year of medical school through the fourth year. Three examples are:
-
Albert Einstein College of Medicine integrates conflict-of-interest education into its first-year medical school curriculum. Students learn about real cases as a way to recognize the existence of conflicts of interest and to drive their reasoning and decision-making skills.
-
The University of Iowa Carver College of Medicine has a third-year medical student course, “Clinical Beginnings,” that is offered before students begin clinical clerkships. The goal is to ensure that students are prepared on how best to handle themselves when they encounter potential conflicts of interest in the field.
-
The Geisel School of Medicine at Dartmouth University has a fourth-year medical student course on clinical pharmacology and therapeutics that incorporates the use of clinical case studies to provide students the tools for being thoughtful and effective prescribers.
* Policy Guide for Academic Medical Centers: Conflict-of-Interest Curriculum Toolkit, Community Catalyst, accessed Nov. 15, 2013, http://www.communitycatalyst.org/doc-store/publications/COI_toolkit_curriculum.pdf.
9. Extension of institutional conflict-of-interest policies to community educational settings
Conflict-of-interest policies established by academic medical centers should apply to all faculty members regardless of the nature of their relationship to the institution—paid or voluntary, full time or part time, on- site or off-site—and to affiliated institutions participating in the academic medical center's educational and training programs.
Conflict-of-interest policies written for medical schools and major teaching hospitals are generally understood to apply to faculty and trainees in those academic settings. However, academic medical centers often affiliate with external community hospitals, clinics, and private practices to provide more learning opportunities for trainees and economic sustenance to the institution.
While there are no systematic data on whether academic medical centers require adjunct or voluntary faculty in community settings to follow conflict-of-interest policies of the “mother institution,” such policies appear to be rare. Obliging such physicians to abide by a stringent set of conflict-of-interest standards may alienate adjunct faculty, putting at risk a network that may be crucial for both patient referrals and outpatient medical training.
For compliance officers, enforcing such policies can be onerous, and certain types of schools may find this policy particularly difficult. New and community-based medical schools, for example, lack strong historical affiliations with hospitals and are especially dependent on the good will of community physicians for their training programs. Similarly, osteopathic medical schools rarely have affiliated teaching hospitals and often provide their entire clinical training within community settings.
Some academic medical centers may choose to adopt policies making it explicit that, while the endpoint is to have their conflict-of-interest policies apply equally to community affiliates and faculty, they may take longer to achieve this goal. Options might include measures such as allowing community sites probationary periods before requiring compliance or permitting exceptions for certain practices with the condition that they provide written management plans for how they will prevent trainees from participating in prohibited industry relationships during rotations.
Example policy: University of California, Los Angeles Medical Center*
“Q: To whom do the new guidelines for relations with industry apply?
A: The new guidelines apply to all faculty (full-time, part-time, or volunteer), staff, trainees (students, residents, and fellows), and volunteers of the David Geffen School of Medicine at UCLA or the UCLA Health. If you have (or should have) a UCLA Health Care or David Geffen School of Medicine at UCLA ID badge, the guidelines apply to you.
Q: Do the guidelines apply to me if I work primarily or exclusively at a UCLA-affiliated hospital or clinic, but not on the UCLA campus?
A: If you are a student, trainee, or faculty rotating to an affiliated program or site away from UCLA, these guidelines apply to you. If you hold a UCLA faculty appointment of any type, these guidelines apply to you regardless of where you work.
If you are a staff member paid through an organization other than UCLA and not working on the Westwood campus, these guidelines may not apply to you. If you are unsure of your status, you may contact [email protected] for clarification.”
* UCLA Health Frequently Asked Questions Regarding Industry Guidelines, UCLA Health, accessed Dec. 2, 2013, http://www.uclahealth.org/body.cfm?id=1189#8.
10. Industry-supported clinical fellowships
In general, clinical fellows, residents, and medical students may not accept industry-sponsored fellowships earmarked specifically for clinical training but may compete for industry fellowships awarded for scientific training. However, scientific fellowships should be awarded under the following circumstances:
-
The fellowship must be primarily related to research training.
-
It must consist of a credible, substantive period of training, rather than, for example, a payment for travel to a conference.
-
It should be awarded based upon a competitive, application-based process.
-
The industry sponsor can have no involvement in review of applicants or selection of awardees.
- The fellowship must not originate from the company's marketing department.
- It must not pay for a residency or clinical fellowship slot.
- Naming of a fellowship after the sponsoring company is discouraged.
- Funding of fellowships by multiple companies should be encouraged.
Industry frequently provides funding for research fellowships, which is appropriate for training the next generation of basic and clinical researchers. This funding should be based on a competitive selection process overseen by a third party (such as an academic medical center or professional medical association), and the industry sponsors should have no involvement in selection.
However, industry grants earmarked specifically for clinical training are potentially more problematic. For example, in 2006 the American Academy of Dermatology, responding to a shortage of specialists in the United States, announced a pilot program in which a group of companies that market dermatologic agents would provide funding to residency programs to support 10 dermatology residency slots at $60,000 per position.41
The program generated debate within the field, both because of concerns that industry sponsors might unduly influence residents and because of disagreements about the severity of the workforce shortage.42 The program was discontinued after the pilot period because of these disagreements.43
More commonly, companies provide support for postgraduate clinical fellowships. For example, the Neurosurgery Research and Education Foundation of the American Association of Neurological Surgeons solicits funding from several medical device companies to provide $50,000 to $75,000 per fellowship for clinical training in areas such as spinal surgery, general neurosurgery, and endovascular neurosurgery. In the 2012-13 academic year, the program sponsored such fellowships at 20 academic medical centers.44
The task force acknowledged that there is as yet no published research on whether industry funding of clinical fellowships leads graduates to favor the sponsoring company's drugs or medical devices. Nonetheless, given the evidence that other forms of commercially funded medical education have led to bias (as outlined in the section on continuing medical education), the task force recommends that academic medical centers resist the temptation to subsidize residencies and clinical fellowships with industry grants. Academic medical centers that choose to accept such funding should ensure that the selection process includes strict safeguards to mitigate industry influence.
11. Ghostwriting and honorary authorship
Academic medical center faculty and trainees should follow the International Committee of Medical Journal Editors standards for authorship and contributorship, which require each author to contribute and participate in the work. This should include significant contributions to conception and design, data collection and analysis, drafting or revising language, and approving final content. Ghostwriting and honorary authorship are strictly prohibited.
Pharmaceutical and device companies have hired medical writing companies to ghostwrite articles that are subsequently published in the medical literature under the names of academics—termed “honorary authors”— who may have had little or no involvement in the research or in the writing. A review of court documents related to Vioxx (rofecoxib), for example, found that clinical trial manuscripts were often authored by Merck employees but attributed to academically affiliated investigators. Only 50 percent of all review articles examined included statements disclosing Merck's sponsorship.45 Another investigation found that Wyeth Pharmaceuticals engaged a medical writing company to produce 26 articles discussing the advantages of its product for hormone replacement therapy, Permarin. The company's funding of these articles was generally undisclosed.46 Physicians rely on the information they read in journal articles to make prescribing decisions, and they should be able to trust that any recommendations made reflect the research and opinions of the authors and not the hidden influence of writers hired by industry.
This recommendation does not apply to situations in which faculty enlist help from professional medical writers employed by their institutions to assist in the drafting of publications. As long as this assistance is acknowledged, it is acceptable and does not constitute ghostwriting.
The International Committee of Medical Journal Editors standards for authorship and contributorship are comprehensive and can guide academic medical centers as they craft their ghostwriting policies.47
Example policy: East Tennessee State University Quillen College of Medicine*
“ETSU faculty, staff, students and trainees are prohibited from having publications or professional presentations of any kind, oral or written, ghostwritten by any party, industry or otherwise. This does not apply to transparent writing collaboration with attribution between academic and industry investigators, medical writers, and/or technical experts.”
* Policy and Guidelines for Interactions Between the James H. Quillen College of Medicine, East Tennessee State University, and Commercial Interests, East Tennessee State University, accessed Nov. 8, 2013, http://www.etsu.edu/com/dean/industry.aspx.
12. Consulting relationships for research and scientific activities
Faculty and trainees should be permitted to engage in consulting relationships with pharmaceutical and device companies about research and scientific matters.
Faculty may provide valuable advice to pharmaceutical and device companies in the service of product innovation or refinement. Examples of such legitimate activities include:
-
Assistance in designing and overseeing clinical trials.
-
Technical assistance in creating or improving medical devices.
-
Advice on potential avenues for future scientific research.
Such consultation opportunities should be spelled out in written contracts with clear deliverables. Compensation must be of fair market value for comparable services.
Example policy: University of Pittsburgh Medical Center*
“In order to avoid gifts disguised as consulting contracts, where SOHS or UPMC personnel have been engaged by Industry to provide consulting services, the consulting contract must provide specific tasks and deliverables, with payment commensurate with the tasks assigned. All such arrangements between individuals or units and outside commercial interests must be reviewed and approved prior to initiation in accordance with appropriate University or UPMC policies.”
* Policy on Conflicts of Interest and Interactions between Representatives of Certain Industries and Faculty, Staff and Students of the Schools of the Health Sciences and Personnel Employed by UPMC at all Domestic Locations,” University of Pittsburgh Medical Center, accessed Nov. 8, 2013, http://www.ogc.pitt.edu/publications/IndustryRelationshipsPolicy.pdf.
13. Consulting relationships for marketing (excluding scientific research and speaking)
Academic medical faculty and trainees should be prohibited from engaging in consulting relationships that are solely or primarily for commercial marketing purposes.
While consultation with industry is a crucial component in drug and device discovery and refinement, some consulting agreements are explicitly focused on marketing. Such consultations vary in topic and scope. For example, advisory boards set up by industry could turn into opportunities for drug representatives to present marketing brochures and seek feedback on the material. Such activities place a physician in a promotional role for a product, which, like serving on industry-sponsored speakers' bureaus, is not consistent with the protection of the integrity of the academic medical center or the authenticity of its educational mission.
Clearly, there can be a fine line between scientific and marketing consultations. A faculty member, for example, may legitimately consult with a company on the clinical utility of a new product and its advantages or disadvantages over competing products. The company may then use this information internally as it creates marketing material. However, this is distinct from an arrangement in which a faculty member is explicitly engaged in the creation or review of promotional material or participating in the development of marketing strategies.
Academic medical centers should develop policies that would prevent faculty from participating in such activities.
Example policy: Stanford University*
“Any consulting agreements with outside entities should carefully delineate and separate your university responsibilities from consulting responsibilities. Specifically, these agreements must not involve or address Stanford University, or its resources and people, including students, postdoctoral scholars and staff. You are responsible for making sure that your consulting activity and the terms of any written agreements are consistent with requirements of the faculty Conflict of Commitment and Interest policy and your university obligations related to inventions and other intellectual property. The School of Medicine also prohibits consulting that is solely or primarily for commercial marketing purposes. To avoid confusion, correspondence and agreements related to consulting activities must not use Stanford letterhead or appear to be Stanford documents. Finally, facilities and services of the University may not be used in connection with your consulting, except in a purely incidental way.”
* Consulting and Other Outside Professional Activities by Members of the Academic Council and Medical Center Line Faculty, Stanford University, accessed Nov. 8, 2013, http://doresearch.stanford.edu/policies/research-policy-handbook/conflicts-commitment-and-interest/consulting-and-other-outside.
14. Pharmaceutical samples
An academic medical center should not accept samples unless it determines that there are compelling circumstances to do so. In these cases, it should implement mechanisms for accepting samples that prevent their use as marketing tools.
While pharmaceutical samples theoretically allow indigent patients to try medications to which they would otherwise not have access, a growing literature has shown that sampling serves primarily as a marketing tactic. Indeed, FDA regulations of sampling under the Prescription Drug Marketing Act of 198748 make it explicit that the agency considers sampling to be a marketing function of companies. Sampling has proved to be an effective marketing technique that tends to unnecessarily drive up health care costs. In one study, resident physicians with access to drug samples were less likely to choose over-the-counter drugs and unadvertised drugs than residents who did not have access to samples.49 In addition, samples are more likely to be given to affluent than poor patients,50 and patients who receive samples ultimately have higher out-of-pocket prescription expenditures than their counterparts who do not receive samples.51 Given this evidence that medication samples serve primarily as marketing tactics for the pharmaceutical industry, academic medical centers should consider either banning their acceptance or use, or setting up safeguards to ensure that samples are used in rational ways that provide long-term benefit to patients.
If samples are permitted, they should be ordered and procured through processes that do not involve interaction with sales representatives. Various state and federal regulations may limit how samples can be received or distributed.
Example policy: Johns Hopkins Medicine*
After a phased-in approach that gave providers and clinics time to adapt to policy changes, Johns Hopkins Medicine stopped accepting samples from industry in 2011. To ensure that underserved patients affected by the ban are able to receive needed medications, the institution created an outpatient medication assistance program for eligible patients.
Its policy states:
“The practice of accepting free pharmaceutical samples risks interference with one's prescribing practices since industry representatives often provide the newest and most costly drugs. Therefore, free pharmaceutical samples and vouchers for free pharmaceutical samples may not be accepted. … When samples are necessary for patient education (e.g., instructing patients in the use of inhalers), they may be accepted, provided they do not carry the name of a company or the name of the company is covered. Johns Hopkins faculty physicians and other hospital staff will educate staff and trainees about and inform patients of affordable options for obtaining medicines. Examples are pharmaceutical assistance programs, vouchers, and large retail chains with low-cost medicines.”
* Johns Hopkins Medicine Policy on Interaction with Industry, Johns Hopkins Medicine, accessed Nov. 8, 2013, http://www.hopkinsmedicine.org/Research/OPC/Policy_Industry_Interaction/policy_interaction_industry.html#section_2d.
15. Pharmacy and therapeutics committee
Ideally, voting members of these committees should not have a financial relationship with industry. In circumstances when this standard cannot be achieved, members with such relationships should be recused from any discussion of, or voting on, a related product, whether the product is manufactured by the company, is a competitor of that product, or is in the same class as that product. All committee members should disclose financial relationships with pharmaceutical and medical device companies, as should practitioners requesting changes or additions to the institution's formulary.
Because pharmacy and therapeutics committees make decisions of profound consequence for both training and patient care, strong measures should be in place to prevent any influence by companies that stand to profit from these decisions. Ideally, academic medical centers should staff their pharmacy and therapeutics committees with qualified faculty who have no financial relationships with drug or device companies. There may be times, however, when an institution determines that the most qualified faculty has such relationships. In that case, to safeguard the integrity of all committee decisions, a member should be recused from discussion or voting when a product comes before the committee that is related to that member's financial interests.
Example policy: Wake Forest Baptist Medical Center*
“Individuals who serve as voting members on Medical Center committees that recommend purchases and utilization of pharmaceutical and medical devices shall recuse themselves from participation in voting and similar decision-making processes when the decision or discussion may pose a real or perceived conflict of interest. In addition, the Chair of the committee may remove a member from the committee in the event the Chair reasonably determines that the member cannot substantially contribute to and participate in the work on the committee due to the member's actual or perceived conflict of interest. Additionally, committee members are required to verbally update any potentially conflicting relationships in open meeting to be documented in the meeting minutes.”
* Wake Forest Baptist Medical Center Policy on Conflict of Commitment and Conflict of Interest, 2012, Wake Forest Baptist Medical Center, accessed Nov. 8, 2013, http://www.amsascorecard.org/institutions/wake-forest-school-of-medicine.
Conflict-of-interest policies at academic medical centers serve to strengthen and protect academic integrity while encouraging those principled relationships with industry that advance research and treatment. The recommendations of this expert task force reflect evolution in the field over the past decade. Many reiterate earlier influential recommendations, including those of the Association of American Medical Colleges and the Institute of Medicine.
Others, such as suggestions regarding site access of pharmaceutical sales representatives and industry funding of continuing medical education, go further, but every recommendation herein is already reflected in the policies of many leading academic medical centers. Although some institutions may encounter resistance from faculty and trainees as they implement policy reforms, progress is possible. Other resources may provide additional guidance to institutional leaders on approaches for successfully developing new policies.52
Ultimately, strong policies will maintain the quality of medical education and support patient trust in the medical profession.
The analysis included in this report is that of The Pew Charitable Trusts and does not necessarily reflect the views of outside reviewers. This report is intended for educational and informative purposes. References to specific policy makers, individuals, schools, policies, associations, or companies have been included solely to advance these purposes and do not constitute an endorsement, sponsorship, or recommendation.
The Pew Charitable Trusts
Susan K. Urahn
Allan Coukell
The Prescription Project
Daniel Carlat, M.D.
Sallyann Bergh
Sarah Carroll
The Expert Task Force on Conflicts of Interest in Medicine
David Korn, M.D. (task force chair), clinical professor of pathology, Harvard Medical School; consultant in pathology, Massachusetts General Hospital; former chief scientific officer, Association of American Medical Colleges; former dean, Stanford University School of Medicine.
Barbara Barnes, M.D., M.S., associate vice chancellor for continuing education and industry relationships, associate dean for continuing medical education, University of Pittsburgh Medical Center.
Daniel Carlat, M.D., director, prescription project, The Pew Charitable Trusts, Washington.
Guy Chisolm, Ph.D., director, Innovation Management and Conflict of Interest Program, Cleveland Clinic.
Marcia Hams, M.A., director, prescription access and quality, Community Catalyst, Boston.
Sharon Levine, M.D., associate executive medical director, Kaiser Permanente, The Permanente Medical Group Inc., Oakland, CA.
Philip A. Pizzo, M.D., professor of pediatrics and microbiology and immunology, immediate past dean, Stanford University School of Medicine.
Reshma Ramachandran, M.D. candidate, Warren Alpert School of Medicine at Brown University; former PharmFree fellow, American Medical Student Association.
John Randolph, J.D., vice president and chief compliance officer, UMass Memorial Healthcare.
Jean Silver-Isenstadt, M.D., Ph.D., executive director, National Physicians Alliance.
James Woolliscroft, M.D., dean, Lyle C. Roll professor of medicine, University of Michigan Medical School.
Heather Pierce, J.D., M.P.H., senior director, Science Policy and Regulatory Counsel, Association of American Medical Colleges, represented the Association of American Medical Colleges in Washington for assistance in providing context to the task force; however, the AAMC has not formally endorsed these recommendations.
Acknowledgments
The project team would like to thank our Pew colleagues—Gaye Williams, Laurie Boeder, Ed Paisley, Lisa Gonzales, Dan Benderly, Bailey Farnsworth, Stephen Howard, Katye Martens, Jerry Tyson, Lisa Plotkin, Carol Hutchinson, and Ben Orlando—for their assistance in readying this report for publication.
This work was made possible by a grant from the Attorney General Consumer and Prescriber Education Grant Program, a fund established by the multistate settlement of consumer fraud claims regarding the marketing of the prescription drug Neurontin. More information on the work being funded by this grant can be found on our website.
The Pew Prescription Project seeks to ensure transparency in physician-industry relationships and promotes policies to reduce or manage conflicts of interest that could affect patient care. Our efforts have supported the creation of new policies at medical schools, professional medical associations, and have successfully supported state and federal disclosure laws.
REFERENCES
1 Office of Inspector General (2003), OIG Compliance Program Guidance for Pharmaceutical Manufacturers, Fed. Reg. 68: 23731-23743.
2 U.S. Senate Committee on Finance, Use of Educational Grants by Pharmaceutical Manufacturers, 110th Cong., 1st sess., (Washington: U.S. Government Printing Office, 2007).
3 Michael A. Steinman, Lisa A. Bero, Mary-Margaret Chren, C. Seth Landefeld, “Narrative Review: The Promotion of Gabapentin: An Analysis of Internal Industry Documents,” Annals of Internal Medicine 145 (2006): 284-293.
4 “Code on Interactions With Health Care Professionals,” Pharmaceutical Research and Manufacturers of America, accessed March 4, 2013, http://www.phrma.org/code-on-interactions-with-healthcare-professionals.
5 “Conflicts of Interest Overview,” Institute on Medicine as a Profession, accessed March 4, 2013, http://www.imapny.org/conflicts_of_interest/conflicts-of-interest-overview.
6 Susan Chimonas, Lisa Patterson, Victoria H. Raveis, David J. Rothman, “Managing Conflicts of Interest in Clinical Care: A National Survey of Policies at U.S. Medical Schools,” Academic Medicine: Journal of the Association of American Medical Colleges 86 (2011): 293-99, doi:10.1097/ACM.0b013e3182a2e204. Susan Chimonas, Susanna D. Evarts, Sarah K. Littlehale, David J. Rothman, “Managing Conflicts of Interest in Clinical Care: The ‘Race to the Middle' at U.S. Medical Schools,” Academic Medicine: Journal of the Association of American Medical Colleges 88, no. 10 (2013): 1464-7, doi: 10.1097/ACM.0b013e3182a2e204.
7 “AMSA PharmFree Scorecard 2013,” American Medical Student Association, accessed Aug. 13, 2013, http://www.amsascorecard.org.
8 AMSA PharmFree Scorecard.
9 Kirsten Austad et al., “Changing Interactions Between Physician Trainees and the Pharmaceutical Industry: A National Survey,” Journal of General Internal Medicine 28, no. 8 (2013): 1064-71, doi:10.1007/s11606-013-2361-0.
10 Ashley Wazana, “Physicians and the Pharmaceutical Industry: Is a Gift Ever Just a Gift?” Journal of the American Medical Association 283, no. 3 (2000): 373-380, doi:10.1001/jama.283.3.373.
11 Jason Dana and George Loewenstein, “A Social Science Perspective on Gifts to Physicians From Industry,” Journal of the American Medical Association 290 (2003): 2, doi:10.1001/jama.290.2.252.
12 Frederick S. Sierles et al., “Medical Students' Exposure to and Attitudes About Drug Company Interactions: A National Survey,” Journal of the American Medical Association 294 (2005): 1034-1042.
13 Samuel M. Keim, Mary Z. Mays, and David Grant, “Interactions Between Emergency Medicine Programs and the Pharmaceutical Industry,” Academic Emergency Medicine 11 (2004): 1, doi:10.1197/j.aem.2003.07.016.
14 C.K. Varley, M.D. Jibson, M. McCarthy, and S. Benjamin, “A Survey of the Interactions Between Psychiatry Residency Programs and the Pharmaceutical Industry,” Academic Psychiatry 29 (2005): 40-46.
15 Accreditation Council for Continuing Medical Education, ACCME Annual Report Data—2007 (2008), http://www.accme.org/sites/default/files/null/Annual_report_2007_20090109.pdf.
16 “AMSA PharmFree Scorecard.”
17 Colleen Walsh, “Medical School Revises Conflict of Interest Policy,” Harvard Gazette, July 21, 2010, http://news.harvard.edu/gazette/story/2010/07/medical-school-revises-conflict-of-interest-policy.
18 Azalea Kim, Lawrence Mumm, and Deborah Korenstein, “ Routine Conflict of Interest Disclosure by Preclinical Lecturers and Medical Students' Attitudes Toward the Pharmaceutical and Device Industries,” Journal of the American Medical Association 308 (2012): 21, doi:10.1001/jama.2012.25315.
19 Federal Food, Drug, and Cosmetic Act, 21 U.S.C. § 502(f); 21 U.S.C. § 352(f); and 21 C.F.R. 201.100(c)(1).
20 “For the Record: Our Work With the U.S. Healthcare and Nonprofit Communities,” GlaxoSmithKline, accessed Nov. 19, 2013, http://fortherecord.payments.us.gsk.com/funding.html.
21 ACCME Annual Report Data 2007.
22 M.A. Bowman and D. L. Pearle, “Changes in Drug Prescribing Patterns Related to Commercial Company Funding of Continuing Medical Education,” Journal of Continuing Education in the Health Professions 8, no. 1 (1988): 13-20.
23 J.P. Orlowski and L. Wateska, “The Effects of Pharmaceutical Firm Enticements on Physician Prescribing Patterns: There's No Such Thing as a Free Lunch,” Chest 102, no. 1 (1992): 270-3.
24 M.E. Dieprink and L. Drogemuller, “Industry-Sponsored Grand Rounds and Prescribing Behavior,” Journal of the American Medical
Association 285 (2001): 1443-44.
25 Harvey P. Katz, Stephen E. Goldfinger, and Suzanne W. Fletcher, “Academia-Industry Collaboration in Continuing Medical Education: Description of Two Approaches,” Journal of Continuing Education in the Health Professions 22(205): 1, doi:10.1002/chp.1340220106.
26 Ronald M. Cervero and Jiange He, “The Relationship Between Commercial Support and Bias in Continuing Medical Education Activities: A Review of the Literature,” Accreditation Council on Continuing Medical Education (2008).
27 Council on Ethical and Judicial Affairs, “Financial Relationships with Industry in Continuing Medical Education,” American Medical Association, 1-A-11, (2011).
28 Association of American Medical Colleges, “The Scientific Basis of Influence and Reciprocity: A Symposium,” 2008.
29 “Sunshine Act Prompts Pharma Companies To Alter Grant Contracts.” The Pink Sheet Daily, October 24, 2013.
30 “UM Medical School Plans Changes to Continuing Medical Education Funding,” University of Michigan Medical School (2010), accessed
Nov. 15, 2013, http://www.uofmhealth.org/u-m-medical-school-plans-changes-continuing-medical-education-funding.
31 Steve Willis, associate dean for continuing medical education at East Carolina University Brody School of Medicine, personal communication to Daniel Carlat, 2013.
32 Federal Food, Drug, and Cosmetic Act.
33 Scott H. Podolsky and Jeremy A. Greene, “A Historical Perspective of Pharmaceutical Promotion and Physician Education,” Journal of the American Medical Association 300 (2008): 7, doi:10.1001/jama.300.7.831.
34 Geoffry Spurling, et al., “Information from Pharmaceutical Companies and the Quality, Quantity, and Cost of Physicians' Prescribing: A Systematic Review,” PLOS Medicine 7 (2010): 10, doi:10.1371/journal.pmed.1000352.
35 B. Mintzes, et al., “Pharmaceutical Sales Representatives and Patient Safety: A Comparative Prospective Study of Information Quality in Canada, France and the United States,” Journal of General Internal Medicine 10: 1368-75, doi:10.1007/s11606-013-2411-7.
36 Alex Berenson, “Eli Lilly Said to Play Down Risk of Top Pill,” New York Times, Dec. 17, 2006, http://www.nytimes.com/2006/12/17/business/17drug.html?_r=0.
37 T. Isaac, J. Zheng, and A. Jha, “Use of UpToDate and Outcomes in U.S. Hospitals,” Journal of Hospital Medicine 7 (2012): 2, doi:10.1002/jhm.944.
38 A.E. Carroll, R.C. Vreeman, J. Buddenbaum, T.S. Inui, “To What Extent Do Educational Interventions Impact Medical Trainees' Attitudes and Behaviors Regarding Industry-Trainee and Industry-Physician Relationships?” Pediatrics 120 (2007): 6, e1528-1535.
39 “Financial Conflict of Interest,” National Institutes of Health, accessed Jan. 14, 2013, http://grants.nih.gov/grants/policy/coi.
40 “AMSA PharmFree Scorecard.”
41 Myrle Croasdale, “Drug Firms to Fund Residency Slots in Dermatology Pilot Program,” American Medical News, July 18, 2005, http://www.amednews.com/article/20050718/profession/307189971/2.
42 Bridget M. Kuehn, “Pharmaceutical Industry Funding for Residencies Sparks Controversy,” Journal of the American Medical Association 293(2005): 13, doi:10.1001/jama.293.13.1572.
43 Alfred Lane M.D., professor of dermatology and pediatrics, Stanford University School of Medicine, and member of the committee of the American Academy of Dermatology that selected residents for the pilot program in 2006, personal communication to Daniel Carlat, Nov. 19, 2013.
44 “NREF Post-Residency Clinical Fellowship Program,” American Association of Neurological Surgeons, accessed Aug. 30, 2013, http://www.aans.org/Grants%20and%20Fellowships/NREF%20Post-Residency%20Clinical%20Fellowship.aspx.
45 Joseph S. Ross, et al., “Guest Authorship and Ghostwriting in Publications Related to Rofecoxib: A Case Study of Industry Documents from Rofecoxib Litigation,” Journal of the American Medical Association 299 (2008): 15, doi:10.1001/jama.299.15.1800.
46 Adriane Fugh-Berman,”The Haunting of Medical Journals: How Ghostwriting Sold ‘HRT,' ” PLOS Medicine 7 (2010): 9, doi:10.1371/journal. pmed.1000335.
47 “Uniform Requirements for Manuscripts Submitted to Biomedical Journals: Ethical Considerations in the Conduct and Reporting of Research: Authorship and Contributorship,” International Committee of Medical Journal Editors, accessed Aug. 30, 2013, http://www.icmje.org/ethical_1author.html.
48 Proclamation No. 92N-0434, 62 Fed. Reg. 64093-64100 (Dec. 3, 1997); and U.S. Food and Drug Administration, The Prescription and Drug Marketing Act (1987), http://www.fda.gov/RegulatoryInformation/Legislation/FederalFoodDrugandCosmeticActFDCAct/SignificantAmendmentstotheFDCAct/PrescriptionDrugMarketingActof1987/.
49 R.F. Adair and L.R. Holmgren, “Do Drug Samples Influence Resident Prescribing Behavior? A Randomized Trial,” American Journal of Medicine 118 (2005): 881-84.
50 Sarah L. Cutrona, et al., “Characteristics of Recipients of Free Prescription Drug Samples: A Nationally Representative Analysis,” American
Journal of Public Health 98(2008): 2, doi:10.2105/AJPH.2007.114249.
51 G.C. Alexander, J. Zhang, and A. Basu, “Characteristics of Patients Receiving Pharmaceutical Samples and Association Between Sample Receipt and Out-of-Pocket Prescription Costs,” Medical Care 46 (2008): 4, doi:10.1097/MLR.0b013e3181618ee0.
52 D.L. Coleman, et al., “Guidelines for Interactions Between Clinical Faculty and the Pharmaceutical Industry: One Medical School's Approach,” [In eng]. Acad Med 81, no. 2 (2008): 154-60. Community Catalyst, “Conflict of Interest Policy Guide for Medical Schools and Academic Medical Centers,” accessed December 2, 2013, http://www.communitycatalyst.org/initiatives-and-issues/initiatives/prescription-reform/conflict-of-interest-policy-guide.






